
Interactions | Reactions | Processes
Classified as: Hydrogenation, catalytic
Catalytic hydrogenation is a multistep process which results in the addition of hydrogen to an organic function.
The first step involves hydrogen, an s-HOMO Lewis base, complexing with a transition metal coated inert support. (Pearson pointed out that bulk metals have Lewis acid properties thus it is not surprising that hydrogen gas should directly react with the metal.) The next step involves the organic moiety also complexing with the metal surface. The process continues with single electron transfer (SET) steps in which the metal complexed hydrogen atoms transfer from an association with the metal to an association with the organic function. Finally the reduced organic function leaves the metal surface which allows the process repeats itself. Note that (1) the reaction is catalytic in the transition metal and (2) the exact mode of reduction is very sensitive to the transition metal used. For more information look in the Chemogenesis webbook sections on redox chemistry and the complexation of hydrogen with metals.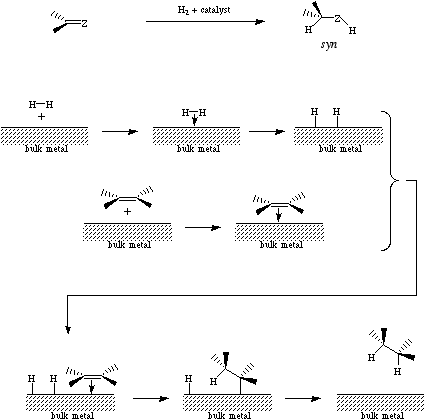
RCOCl |
+ |
H2 |
+ |
Pd on BaSO4 |
RCHO |
+ |
HCl |
+ |
H2 |
+ |
Ni/H2 Raney Nickel |
R–CH3 |
+ |
H2 |
+ |
Pd on C |
R–CH3 |
R–C≡C–R |
+ |
H2 |
+ |
Pd + Pb on CaCO3 Lindlar Cat. |
+ |
2 |
H2 |
+ |
Pd on C |
+ |
H2O |
+ |
H2 |
+ |
Ni/H2 Raney Nickel |
+ |
H2 |
+ |
Pt |
R–C≡C–R |
+ |
H2 |
+ |
Pd on C |
R–C≡C–R |
+ |
2 |
H2 |
+ |
Pd on C |
R–C≡C–R |
+ |
2 |
H2 |
+ |
Pt on carbon |
+ |
H2 |
+ |
Pd on C |
+ |
H2 |
+ |
Rh on Carbon |
RCOOH |
+ |
2 |
H2 |
+ |
Ru on carbon |
RCH2OH |
+ |
2 |
H2O |
RCHO |
+ |
H2 |
+ |
Ru on carbon |
+ |
H2O |
RCH2OH |
+ |
H2 |
+ |
Pd on C |
+ |
2 |
H2 |
+ |
Pd on C |
+ |
H2O |
+ |
H2 |
+ |
Ru on carbon |
+ |
3 |
H2 |
+ |
Rh on Carbon |
2 |
+ |
6 |
H2 |
+ |
Pd on C |
+ |
NH3 |
+ |
2 |
H2 |
+ |
Pd on C |
+ |
H+(aq) |
+ |
R–NH2 |
+ |
3 |
H2 |
+ |
Rh on Carbon |
+ |
H2 |
+ |
Pd on C |
+ |
RCl |
+ |
H2 |
+ |
Pd on C |
R–H |
+ |
HCl |
RBr |
+ |
H2 |
+ |
Pd on C |
R–H |
+ |
HBr |
R–I |
+ |
H2 |
+ |
Pd on C |
R–H |
+ |
HI |
+ |
H2 |
+ |
Pd on C |
R–CH2CH2OH |
RHC=N–NH2 |
+ |
H2 |
+ |
Pt on carbon |
R–CH2–NNH2 |
+ |
H2 |
+ |
Pt on carbon |
+ |
H2 |
+ |
Ru on carbon |
+ |
H2O |
R2CHOH |
+ |
H2 |
+ |
Pd on C |
+ |
4 |
H2 |
+ |
Rh on Carbon |
R–CN |
+ |
2 |
H2 |
+ |
Pd on C |
RCH2NH2 |
2 |
R–CN |
+ |
4 |
H2 |
+ |
Rh on Carbon |
(RCH2)2NH |
+ |
NH3 |
R–CN |
+ |
R–NH2 |
+ |
4 |
H2 |
+ |
Rh on Carbon |
+ |
NH3 |
3 |
R–CN |
+ |
6 |
H2 |
+ |
Pd on C |
2 |
(RCH2)3N |
+ |
2 |
NH3 |
+ |
2 |
H2 |
+ |
Pd on C |
2 |
+ |
4 |
H2 |
+ |
Pt on carbon |
+ |
NH3 |
+ |
H2 |
+ |
H2O |
+ |
Pd on C |
+ |
H+(aq) |
+ |
NH3 |
+ |
3 |
H2 |
+ |
Ni |
+ |
2 |
H2O |
+ |
2 |
H2 |
+ |
Pt on carbon |
+ |
CH3CH2OH |
+ |
H2O |
+ |
2 |
H2 |
+ |
Pt on carbon |
+ |
H2O |
R2N–NO |
+ |
2 |
H2 |
+ |
Pd on C |
(R)2N–NH2 |
+ |
H2O |
+ |
2 |
H2 |
+ |
Pd on C |
+ |
H2O |
+ |
3 |
H2 |
+ |
Rh on Carbon |
+ |
H2 |
+ |
Pd on C |
+ |
H2O |
+ |
+ |
H2 |
+ |
Pt on carbon |
+ |
H2O |
+ |
2 |
H2 |
+ |
Pd on C |
+ |
H2 |
+ |
Pd on C |
+ |
H2 |
+ |
Pd on C |
+ |
(CH3)3COH |
+ |
+ |
H2 |
+ |
Pd on C |
+ |
+ |
+ |
H2 |
+ |
Pd on C |
+ |
+ |
H2 |
+ |
Pd on C |
+ |
H2 |
R–NO2 |
+ |
3 |
H2 |
+ |
Pd on C |
R–NH2 |
+ |
2 |
H2O |
+ |
H2 |
+ |
Pd on C |
+ |
R–OH |
+ |
2 |
H2 |
+ |
Pd on C |
RCH2OH |
+ |
R–OH |
+ |
2 |
H2 |
+ |
Pd on C |
+ |
H2O |
HC≡CH |
+ |
H2 |
H2C=CH2 |
+ |
H2 |
+ |
Ni/H2 Raney Nickel |
+ |
2 |
H2 |
+ |
Ni/H2 Raney Nickel |
+ |
5 |
H2 |
+ |
Ni |
+ |
H2O |
+ |
H2 |
+ |
Ni |
2 |
CH3CHO |
+ |
H2 |
+ |
H2 |
+ |
2 |
NaOH |
+ |
2 |
NaCl |
+ |
H2 |
+ |
H2 |
+ |
H2 |
+ |
H2 |
+ |
H2 |
+ |
2 |
H2 |
+ |
2 |
H2 |
+ |
Pd |
+ |
3 |
H2 |
+ |
Pd on C |
+ |
H2 |
+ |
H2O |
+ |
H2 |
+ |
+ |
4 |
+ |
14 |
H2 |
+ |
Ni |
+ |
+ |
+ |
+ |
4 |
H2O |
+ |
HNO3|H2SO4 concentrated |
+ |
H2 |
+ |
2 |
+ |
2 |
HCN |
+ |
2 |
H2 |
+ |
Pd on BaSO4 |
+ |
2 |
H2O |
+ |
2 |
+ |
2 |
HCN |
+ |
2 |
H2 |
+ |
Pd on BaSO4 |
+ |
2 |
H2O |
+ |
2 |
+ |
2 |
HCN |
+ |
2 |
H2 |
+ |
Pd on BaSO4 |
+ |
2 |
H2O |
+ |
2 |
+ |
2 |
HCN |
+ |
2 |
H2 |
+ |
Pd on BaSO4 |
+ |
2 |
H2O |
+ |
2 |
+ |
2 |
HCN |
+ |
2 |
H2 |
+ |
Pd on BaSO4 |
+ |
2 |
H2O |
+ |
2 |
+ |
2 |
HCN |
+ |
2 |
H2 |
+ |
Pd on BaSO4 |
+ |
2 |
H2O |
+ |
2 |
+ |
2 |
HCN |
+ |
2 |
H2 |
+ |
Pd on BaSO4 |
+ |
2 |
H2O |
+ |
+ |
H2 |
+ |
H2O |
+ |
4 |
H2 |
+ |
Ni |
+ |
3 |
H2 |
+ |
Ni |
+ |
2 |
H2O |
Sunflower Oil |
+ |
H2 |
+ |
Ni |
Soft Margarine |
Sunflower Oil |
+ |
H2 |
+ |
Ni |
Hard Margarine |
+ |
H2 |
+ |
Ni |
+ |
H2 |
+ |
Ni |
CH3CN |
+ |
2 |
H2 |
+ |
Ni |
CH3CH2NH2 |
+ |
H2 |
+ |
Ni |
+ |
H2 |
+ |
Pd |
+ |
2 |
H2 |
+ |
Ni |
© Mark R. Leach 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com