
Interactions | Reactions | Processes
Classified as: Decomposition by Heat (Pyrolysis)
On heating compounds may react so that they lose gaseous components.
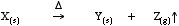
+ |
CH3• |
+ |
Pt |
+ |
3 |
H2 |
KCl |
Na2CO3 |
CuCO3 (unknown) |
CuO |
+ |
CO2 |
2 |
NaNO3 |
2 |
NaNO2 |
+ |
O2 |
2 |
Pb(NO3)2 |
2 |
PbO |
+ |
O2 |
+ |
4 |
NO2 |
CuSO4.5H2O |
CuO |
+ |
SO3 |
+ |
5 |
H2O |
Cu(OH)2 |
+ |
OH–(aq) |
CuO |
+ |
H2O |
6 |
CH4 |
+ |
O2 |
2 |
HC≡CH |
+ |
2 |
CO |
+ |
10 |
H2 |
+ |
CO2 |
+ |
CO |
+ |
H2O |
2 |
HgO |
2 |
Hg |
+ |
O2 |
2 |
CuO |
+ |
3 |
Cu |
+ |
SO2 |
2 |
NaHCO3 |
Na2CO3 |
+ |
CO2 |
+ |
H2O |
2 |
KHCO3 |
K2CO3 |
+ |
CO2 |
+ |
H2O |
4 |
NaNO3 |
2 |
Na2O |
+ |
5 |
O2 |
+ |
2 |
N2 |
4 |
NaNO2 |
2 |
Na2O |
+ |
3 |
O2 |
+ |
2 |
N2 |
BeCO3 |
BeO |
+ |
CO2 |
MgCO3 |
MgO |
+ |
CO2 |
CaCO3 |
CaO |
+ |
CO2 |
SrCO3 |
SrO |
+ |
CO2 |
BaCO3 |
BaO |
+ |
CO2 |
CaSO4 .2H2O |
CaSO4 .½H2O |
+ |
1.5 |
H2O |
CaSO4 .½H2O |
CaSO4 |
+ |
H2O |
CaSO4 |
CaO |
+ |
SO3 |
2 |
LiOH |
Li2O |
+ |
H2O |
BeSO4 |
BeO |
+ |
SO3 |
MgSO4 |
MgO |
+ |
SO3 |
SrSO4 |
SrO |
+ |
SO3 |
BaSO4 |
BaO |
+ |
SO3 |
2 |
BI3 |
2 |
B |
+ |
3 |
I2 |
B2H6 |
2 |
B |
+ |
3 |
H2 |
Na[Al(OH)4] |
NaAlO2 |
+ |
2 |
H2O |
H3BO3 |
HBO2 |
+ |
H2O |
2 |
HBO2 |
B2O3 |
+ |
H2O |
2 |
H3BO3 |
B2O3 |
+ |
3 |
H2O |
+ |
H2O |
NH4NO3 |
N2 |
+ |
2 |
H2O |
2 |
NaN3 |
3 |
N2 |
+ |
2 |
Na |
4 |
FeS2.FeAs2 |
As4 |
+ |
4 |
FeS |
NH4Cl |
NH3 |
+ |
HCl |
NH4NO3 |
N2O |
+ |
2 |
H2O |
2 |
NH4NO3 |
2 |
N2 |
+ |
O2 |
+ |
4 |
H2O |
4 |
H2N–OH |
NH3 |
+ |
N2 |
+ |
HNO2 |
+ |
NO2 |
2 |
HN3 |
H2 |
+ |
3 |
N2 |
2 |
HNO2 |
NO2 |
+ |
NO |
+ |
H2O |
N2O5 |
NO2 |
+ |
NO |
+ |
O2 |
Sb2O5 |
Sb2O3 |
+ |
O2 |
As2O5 |
As2O3 |
+ |
O2 |
2 |
H3PO4 |
+ |
H2O |
2 |
O3 |
3 |
O2 |
2 |
2 |
H2SO4 |
+ |
O2 |
Te |
+ |
4 |
HNO3 |
TeO2 |
+ |
2 |
H2O |
+ |
4 |
NO2 |
S8 |
4 |
S2 |
Ag2CO3 |
Ag2O |
+ |
CO2 |
2 |
HOOH |
2 |
H2O |
+ |
O2 |
2 |
SeBr4 |
S2Br2 |
+ |
3 |
Br2 |
2 |
HIO3 |
I2O5 |
+ |
H2O |
2 |
HOF |
2 |
HF |
+ |
O2 |
2 |
KClO3 |
2 |
KCl |
+ |
3 |
O2 |
KClO4 |
KCl |
+ |
2 |
O2 |
H5IO6 |
HIO4 |
+ |
2 |
H2O |
2 |
HIO4 |
I2O5 |
+ |
O2 |
+ |
H2O |
I2Cl6 |
2 |
I–Cl |
+ |
2 |
Cl2 |
P |
2 |
P2 |
P |
4 |
P Red |
2 |
H2C=CH2 |
N≡C–N3 |
N≡C–N |
+ |
N2 |
CH3CH3 |
H2C=CH2 |
+ |
H2 |
2 |
2 |
R–C–R |
+ |
2 |
[NH4]+ |
+ |
NH3 |
+ |
2 |
H2O |
+ |
Pt |
+ |
4 |
H2 |
Coal |
Coke |
+ |
CO + H2 |
+ |
Coal Tar |
CH3COOH |
+ |
(CH3CH2O)3P=O |
H2C=C=O |
+ |
H2O |
+ |
H2 |
Ca(HCO3)2 |
+ |
H2O |
CaCO3 |
+ |
CO2 |
+ |
H2O |
2 |
FeCl3 |
2 |
FeCl2 |
+ |
Cl2 |
Na2[B4O5(OH)4] .8H2O |
Na2B4O7 |
+ |
10 |
H2O |
HC≡CH |
2 |
C |
+ |
H2 |
FeS2 pyrite |
S |
+ |
FeS |
4 |
FeS2 pyrite |
+ |
11 |
O2 |
2 |
Fe2O3 |
+ |
8 |
SO2 |
2 |
H2SO4 |
2 |
SO2 |
+ |
O2 |
+ |
2 |
H2O |
H2S2O7 |
SO3 |
+ |
H2SO4 |
SiF4 |
+ |
2 |
H2O |
SiO2 |
+ |
4 |
HF |
2 |
Al(OH)3 |
Al2O3 |
+ |
3 |
H2O |
2 |
+ |
NH3 |
75% KNO3, 15% S, 10% C gunpowder |
N2 |
+ |
CO2 |
+ |
K2S |
+ |
NO2 |
NaCl |
+ |
NH3 |
+ |
CO2 |
+ |
H2O |
NaHCO3 |
+ |
NH4Cl |
4 |
FeCr2O4 |
+ |
8 |
Na2CO3 |
+ |
7 |
O2 |
8 |
Na2CrO4 |
+ |
2 |
Fe2O3 |
+ |
8 |
CO2 |
SiHCl3 |
+ |
H2 |
Si |
+ |
3 |
HCl |
Carbon Fiber |
2 |
BCl3 |
+ |
3 |
H2 |
2 |
Boron Fiber |
+ |
6 |
HCl |
CH3SiCl3 |
SiC Fiber |
+ |
3 |
HCl |
3 |
SiO2 |
+ |
6 |
C |
+ |
2 |
N2 |
Si3N4 |
+ |
6 |
CO |
CO2 |
+ |
N2 |
+ |
H2O |
+ |
NO2 |
HCONH2 |
HCN |
+ |
H2O |
+ |
+ |
H2C=CH2 |
+ |
CH3–CH=CH2 |
Gas Oil |
+ |
Gasoline |
+ |
Naphthene |
Gas Oil |
+ |
LPG |
+ |
H2 |
+ |
+ |
Gasoline |
+ |
Naphtha |
CH4 |
+ |
Natural Gas |
+ |
Naphtha |
+ |
Crude Oil |
HC≡CH |
+ |
H2C=CH2 |
+ |
+ |
+ |
+ |
H2 |
+ |
H2O |
+ |
HCHO |
+ |
H2O |
CH3CH2OH |
+ |
Cu |
CH3CHO |
+ |
H2 |
+ |
H+ |
+ |
H2O |
Pb(N3)2 |
+ |
3 |
N2 |
PCl5 |
PCl3 |
+ |
Cl2 |
2 |
+ |
+ |
2 |
H2O |
2 |
NO2 |
2 |
NO |
+ |
O2 |
FeC2O4 |
FeO |
+ |
CO2 |
+ |
CO |
+ |
+ |
[CH3CH2]+ |
+ |
[CH3]+ |
3 |
+ |
3 |
O2 |
CH3COONa |
+ |
NaOH |
CH4 |
+ |
Na2CO3 |
© Mark R. Leach 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com