
Interactions | Reactions | Processes
Classified as: Hydrolysis
The term hydrolysis means "breaking-up" (lysis) "due to water" (hydro).
Many reactions are of the form X-Y + H2O -> X-H + HO-Y, and these reactions can be driven by removing water. Hydrolysis reactions are usually multi-step processes with sequences of protonation followed by attack of HO- or H2O, or attack of HO- followed by protonation.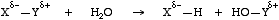
+ |
H2O |
+ |
H+(aq) |
RCHO |
+ |
2 |
R–OH |
(CH3CO)2CO |
+ |
2 |
[OH]– |
2 |
[CH3COO]– |
+ |
H2O |
RCOCl |
+ |
H2O |
RCOOH |
+ |
HCl |
+ |
H+(aq) |
[BH4]– |
+ |
4 |
H2O |
4 |
H2 |
+ |
[B(OH)4]– |
[AlH4]– |
+ |
4 |
H2O |
4 |
H2 |
+ |
[Al(OH)4]– |
+ |
H2O |
R–OH |
+ |
HCHO |
+ |
H+ |
+ |
H2O |
HCOOR |
+ |
R–OH |
+ |
H+ |
B2H6 |
+ |
8 |
H2O |
2 |
[B(OH)4]– |
+ |
6 |
H2 |
+ |
2 |
H+ |
+ |
H2 |
+ |
H2O |
+ |
Pd on C |
+ |
H+(aq) |
+ |
NH3 |
+ |
2 |
Li |
+ |
CH3CH2NH2 |
+ |
2 |
[H] |
+ |
H2O |
+ |
CH3OH |
+ |
2 |
Li+ |
+ |
H2O |
+ |
H+(aq) |
+ |
CH3OH |
+ |
H2O |
+ |
(CH3)2NH |
+ |
H2O |
+ |
H+ |
RCHO |
+ |
CH3COOR |
+ |
H2O |
+ |
OH–(aq) |
[CH3COO]– |
+ |
R–OH |
CH3COOR |
+ |
H2O |
+ |
H+(aq) |
CH3COOH(aq) |
+ |
R–OH |
+ |
H2O |
RLi |
+ |
H2O |
R–H |
+ |
Li+ |
+ |
[OH]– |
RSO2Cl |
+ |
H2O |
R–SO3H |
+ |
HCl |
+ |
H2O |
+ |
H+(aq) |
+ |
RCOOH |
+ |
2 |
NaOH |
+ |
OH–(aq) |
+ |
RCOO– |
+ |
2 |
Na+ |
+ |
NaOH |
+ |
OH–(aq) |
RCOO– |
+ |
CH3OH |
+ |
H2O |
+ |
Li+ |
+ |
[OH]– |
+ |
H2O |
+ |
H+(aq) |
+ |
CH3COOH |
[BH4]– |
+ |
H+ |
+ |
3 |
H2O |
H3BO3 |
+ |
4 |
H2 |
CaNCN |
+ |
3 |
H2O |
2 |
NH3 |
+ |
CaCO3 |
Li3N |
+ |
3 |
H2O |
3 |
LiOH |
+ |
NH3 |
Li2O |
+ |
H2O |
2 |
LiOH |
Na2O |
+ |
H2O |
2 |
NaOH |
KO2 |
+ |
H2O |
2 |
KOH |
2 |
NaNO2 |
+ |
2 |
HCl |
+ |
H2O |
NO2 |
+ |
NO |
+ |
2 |
NaCl |
+ |
H2O |
2 |
[NO2]– |
+ |
2 |
H+ |
+ |
H2O |
NO2 |
+ |
NO |
+ |
H2O |
LiH |
+ |
H2O |
LiOH |
+ |
H2 |
NaH |
+ |
H2O |
NaOH |
+ |
H2 |
KH |
+ |
H2O |
KOH |
+ |
H2 |
RbH |
+ |
H2O |
RbOH |
+ |
H2 |
CsH |
+ |
H2O |
CsOH |
+ |
H2 |
Rb2O |
+ |
H2O |
2 |
RbOH |
Cs2O |
+ |
H2O |
2 |
CsOH |
LiC≡CLi |
+ |
2 |
H2O |
HC≡CH |
+ |
2 |
LiOH |
Be3N2 |
+ |
6 |
H2O |
3 |
Be(OH)2 |
+ |
2 |
NH3 |
Mg3N2 |
+ |
6 |
H2O |
3 |
Mg(OH)2 |
+ |
2 |
NH3 |
Ca3N2 |
+ |
6 |
H2O |
3 |
Ca(OH)2 |
+ |
2 |
NH3 |
Sr3N2 |
+ |
6 |
H2O |
3 |
Sr(OH)2 |
+ |
2 |
NH3 |
Ba3N2 |
+ |
6 |
H2O |
3 |
Ba(OH)2 |
+ |
2 |
NH3 |
BeH2 |
+ |
2 |
H2O |
Be(OH)2 |
+ |
2 |
H2 |
MgH2 |
+ |
2 |
H2O |
Mg(OH)2 |
+ |
2 |
H2 |
CaH2 |
+ |
2 |
H2O |
Ca(OH)2 |
+ |
2 |
H2 |
SrH2 |
+ |
2 |
H2O |
Sr(OH)2 |
+ |
2 |
H2 |
BaH2 |
+ |
2 |
H2O |
Ba(OH)2 |
+ |
2 |
H2 |
Be2C |
+ |
4 |
H2O |
2 |
Be(OH)2 |
+ |
CH4 |
MgC2 |
+ |
2 |
H2O |
HC≡CH |
+ |
Mg(OH)2 |
SrC2 |
+ |
2 |
H2O |
HC≡CH |
+ |
Sr(OH)2 |
BaC2 |
+ |
2 |
H2O |
HC≡CH |
+ |
Ba(OH)2 |
4 |
BF3 |
+ |
3 |
H2O |
H3BO3 |
+ |
3 |
H+ |
+ |
3 |
[BF4]– |
BBr3 |
+ |
3 |
H2O |
H3BO3 |
+ |
3 |
HBr |
BCl3 |
+ |
3 |
H2O |
H3BO3 |
+ |
3 |
HCl |
BI3 |
+ |
3 |
H2O |
H3BO3 |
+ |
3 |
HI |
B2H6 |
+ |
6 |
H2O |
2 |
H3BO3 |
+ |
6 |
H2 |
+ |
9 |
H2O |
3 |
H3BO3 |
+ |
3 |
NH3 |
+ |
3 |
H2 |
+ |
H2O |
2 |
HCl |
+ |
CO2 |
CCl4 |
+ |
H2O |
+ |
2 |
HCl |
Ca3P2 |
+ |
6 |
H2O |
2 |
PH3 |
+ |
3 |
Ca(OH)2 |
P |
+ |
3 |
NaOH |
+ |
3 |
H2O |
PH3 |
+ |
3 |
Na+ |
+ |
3 |
[H2PO2]– |
Cs3As |
+ |
3 |
H2O |
H3As |
+ |
3 |
CsOH |
K3As |
+ |
3 |
H2O |
H3As |
+ |
3 |
KOH |
Li3As |
+ |
3 |
H2O |
H3As |
+ |
3 |
LiOH |
Na3As |
+ |
3 |
H2O |
H3As |
+ |
3 |
NaOH |
Rb3As |
+ |
3 |
H2O |
H3As |
+ |
3 |
RbOH |
Cs3Sb |
+ |
3 |
H2O |
H3Sb |
+ |
3 |
CsOH |
K3Sb |
+ |
3 |
H2O |
H3Sb |
+ |
3 |
KOH |
Li3Sb |
+ |
3 |
H2O |
H3Sb |
+ |
3 |
LiOH |
Na3Sb |
+ |
3 |
H2O |
H3Sb |
+ |
3 |
NaOH |
Rb3Sb |
+ |
3 |
H2O |
H3Sb |
+ |
3 |
RbOH |
NCl3 |
+ |
4 |
H2O |
[NH4]+ |
+ |
[OH]– |
+ |
3 |
HClO |
PCl3 |
+ |
3 |
H2O |
H3PO3 |
+ |
3 |
HCl |
PCl5 |
+ |
4 |
H2O |
H3PO4 |
+ |
5 |
HCl |
PCl5 |
+ |
H2O |
POCl3 |
+ |
2 |
HCl |
N2O4 |
+ |
H2O |
HNO3 |
+ |
HNO2 |
3 |
NO2 |
+ |
H2O |
2 |
HNO3 |
+ |
NO |
N2O5 |
+ |
H2O |
2 |
HNO3 |
2 |
P2O3 |
+ |
6 |
H2O |
4 |
H3PO3 |
2 |
H3PO4 |
+ |
H2O |
+ |
H2O |
H3PO4 |
+ |
H3PO3 |
P |
+ |
3 |
[OH]– |
+ |
3 |
H2O |
PH3 |
+ |
3 |
[H2PO2]– |
Al2O3 |
+ |
6 |
HCl |
2 |
Al3+ |
+ |
6 |
Cl– |
+ |
3 |
H2O |
Al2O3 |
+ |
2 |
NaOH |
+ |
3 |
H2O |
2 |
Na[Al(OH)4] |
Al2O3 |
+ |
2 |
[OH]– |
+ |
3 |
H2O |
2 |
[Al(OH)4]– |
+ |
H2O |
HOOH |
+ |
H2SO4 |
B2O3 |
+ |
3 |
H2O |
2 |
H3BO3 |
N2O3 |
+ |
H2O |
2 |
HNO2 |
Na2SO3 |
+ |
2 |
HCl |
+ |
H2O |
SO2 |
+ |
2 |
NaCl |
+ |
H2O |
2 |
S |
+ |
H2S |
+ |
SO2 |
+ |
H2SO4 |
+ |
H2O |
+ |
H2SO4 |
SOCl2 |
+ |
H2O |
SO2 |
+ |
2 |
HCl |
SOF2 |
+ |
H2O |
SO2 |
+ |
2 |
HF |
SOBr2 |
+ |
H2O |
SO2 |
+ |
2 |
HBr |
TeF6 |
+ |
6 |
H2O |
Te(OH)6 |
+ |
6 |
HF |
SF4 |
+ |
2 |
H2O |
SO2 |
+ |
4 |
HF |
SCl4 |
+ |
2 |
H2O |
SO2 |
+ |
4 |
HCl |
2 |
ClO2 |
+ |
H2O |
+ |
hν |
O2 |
+ |
2 |
H+ |
+ |
[ClO3]– |
+ |
Cl– |
6 |
BrO2 |
+ |
6 |
[OH]– |
5 |
[BrO3]– |
+ |
Br– |
+ |
3 |
H2O |
ClF |
+ |
H2O |
HClO |
+ |
HF |
BrF |
+ |
H2O |
HOBr |
+ |
HF |
BrCl |
+ |
H2O |
HOBr |
+ |
HCl |
I–Cl |
+ |
H2O |
HOI |
+ |
HCl |
I–Br |
+ |
H2O |
HOI |
+ |
HBr |
I–F |
+ |
H2O |
HOI |
+ |
HF |
ClF3 |
+ |
2 |
H2O |
HClO2 |
+ |
3 |
HF |
ClF5 |
+ |
3 |
H2O |
HClO3 |
+ |
5 |
HF |
BrF5 |
+ |
3 |
H2O |
HBrO3 |
+ |
5 |
HF |
IF5 |
+ |
3 |
H2O |
HIO3 |
+ |
5 |
HF |
2 |
XeF2 |
+ |
2 |
H2O |
2 |
Xe |
+ |
4 |
HF |
+ |
O2 |
6 |
XeF4 |
+ |
12 |
H2O |
2 |
XeO3 |
+ |
4 |
Xe |
+ |
24 |
HF |
+ |
3 |
O2 |
XeF6 |
+ |
3 |
H2O |
XeO3 |
+ |
6 |
HF |
XeO3 |
+ |
NaOH |
Na+ |
+ |
[HXeO4]– |
2 |
Na+ |
+ |
2 |
[HXeO4]– |
+ |
2 |
NaOH |
4 |
Na+ |
+ |
[XeO6]4– |
+ |
Xe |
+ |
O2 |
+ |
2 |
H2O |
+ |
H2O |
+ |
HCl(aq) |
+ |
CO2 |
+ |
CH3CH2OH |
+ |
H2O |
+ |
CH3OH |
2 |
+ |
Na2CO3 |
+ |
H2O |
2 |
+ |
CO2 |
+ |
2 |
NaCl |
2 |
+ |
2 |
+ |
6 |
H2O |
4 |
+ |
6 |
CH3COOH |
Na2[B4O5(OH)4] .8H2O |
+ |
2 |
HCl(aq) |
4 |
H3BO3 |
+ |
2 |
NaCl |
+ |
5 |
H2O |
Na2[B4O5(OH)4] .8H2O |
+ |
5 |
H2O |
2 |
H3BO3 |
+ |
2 |
[B(OH)4]– |
+ |
2 |
Na+ |
+ |
8 |
H2O |
Na2 [B2(OH)4(O2)2] .6H2O |
+ |
H2O |
2 |
HOOH |
+ |
2 |
[B(OH)4]– |
+ |
2 |
Na+ |
+ |
2 |
H2O |
+ |
2 |
H2O |
H2N–NH2 |
+ |
2 |
SiF4 |
+ |
2 |
H2O |
SiO2 |
+ |
4 |
HF |
NaOCl(aq) |
+ |
2 |
HCl |
Cl2 |
+ |
NaCl |
+ |
H2O |
XeF6 |
+ |
H2O |
XeOF4 |
+ |
2 |
HF |
1000 |
(CH3)2SiCl2 |
+ |
1000 |
H2O |
[(CH3)2SiO]n |
+ |
2000 |
HCl |
2 |
HCONH2 |
+ |
H2SO4 |
+ |
2 |
H2O |
2 |
HCOOH |
+ |
(NH4)2SO4 |
+ |
H2O |
+ |
HCHO |
+ |
H2O |
+ |
H2O |
+ |
2 |
H2O |
+ |
NH3 |
+ |
H2O |
+ |
NH3 |
(C2H5O)2SO2 |
+ |
2 |
H2O |
+ |
H2SO4 |
2 |
CH3CH2OH |
+ |
+ |
H2SO4 |
+ |
1000 |
NaOH |
+ |
H2O |
+ |
H2SO4 |
+ |
[NH4]+ |
+ |
[HSO4]– |
+ |
CH3OH |
+ |
H2SO4 |
+ |
[NH4]+ |
+ |
[HSO4]– |
+ |
H2SO4 |
+ |
2 |
H2O |
+ |
[NH4]+ |
+ |
[HSO4]– |
+ |
H2SO4 |
+ |
H2O |
+ |
CH3OH |
+ |
[NH4]+ |
+ |
[HSO4]– |
+ |
H2O |
+ |
NaOH |
+ |
NaCl |
+ |
HOOH |
+ |
HOOH |
+ |
H2O |
+ |
H+ |
+ |
+ |
+ |
H2O |
Polyurethane |
+ |
CO2 |
2 |
+ |
2 |
HCN |
+ |
2 |
H2 |
+ |
Pd on BaSO4 |
+ |
2 |
H2O |
+ |
2 |
+ |
2 |
HCN |
+ |
2 |
H2 |
+ |
Pd on BaSO4 |
+ |
2 |
H2O |
+ |
2 |
+ |
2 |
HCN |
+ |
2 |
H2 |
+ |
Pd on BaSO4 |
+ |
2 |
H2O |
+ |
2 |
+ |
2 |
HCN |
+ |
2 |
H2 |
+ |
Pd on BaSO4 |
+ |
2 |
H2O |
+ |
2 |
+ |
2 |
HCN |
+ |
2 |
H2 |
+ |
Pd on BaSO4 |
+ |
2 |
H2O |
+ |
2 |
+ |
2 |
HCN |
+ |
2 |
H2 |
+ |
Pd on BaSO4 |
+ |
2 |
H2O |
+ |
2 |
+ |
2 |
HCN |
+ |
2 |
H2 |
+ |
Pd on BaSO4 |
+ |
2 |
H2O |
+ |
Sucrose |
+ |
H2O |
+ |
H+ |
+ |
Starch |
+ |
1000 |
H2O |
1000 |
Cellulose |
+ |
1000 |
H2O |
+ |
H+ |
1000 |
Butter |
+ |
H2O |
+ |
NaOH(aq) |
+ |
CH3(CH2)14COOH |
+ |
Oleic Acid |
+ |
CH3(CH2)12COOH |
Lard |
+ |
H2O |
+ |
NaOH(aq) |
+ |
Oleic Acid |
+ |
CH3(CH2)14COOH |
+ |
CH3(CH2)16COOH |
Corn Oil |
+ |
H2O |
+ |
NaOH(aq) |
+ |
Oleic Acid |
+ |
Linoleic Acid |
+ |
CH3(CH2)14COOH |
Linseed Oil |
+ |
H2O |
+ |
NaOH(aq) |
+ |
Linolenic Acid |
+ |
Linoleic Acid |
+ |
Oleic Acid |
Olive Oil |
+ |
H2O |
+ |
NaOH(aq) |
+ |
Oleic Acid |
+ |
CH3(CH2)14COOH |
+ |
Linoleic Acid |
Peanut Oil |
+ |
H2O |
+ |
NaOH(aq) |
+ |
Oleic Acid |
+ |
Linoleic Acid |
+ |
CH3(CH2)14COOH |
Coconut Oil |
+ |
H2O |
+ |
NaOH(aq) |
+ |
CH3(CH2)10COOH |
+ |
CH3(CH2)12COOH |
+ |
CH3(CH2)14COOH |
Peanut Oil |
+ |
Coconut Oil |
+ |
KOH |
+ |
H2O |
Soap |
+ |
(CH3CO)2CO |
+ |
H2O |
2 |
CH3COOH |
CH3COCl |
+ |
H2O |
CH3COOH |
+ |
HCl |
AlCl3 |
+ |
6 |
H2O |
[Al(H2O)6]3+ |
+ |
3 |
Cl– |
TiCl4 |
+ |
2 |
H2O |
TiO2 |
+ |
4 |
HCl |
+ |
[OH]– |
+ |
[CH3COO]– |
GeCl4 |
+ |
2 |
H2O |
GeO2 |
+ |
4 |
HCl |
CH3CH2CN |
+ |
2 |
H2O |
+ |
HCl(aq) |
CH3CH2COOH |
+ |
NH3 |
+ |
H2O |
+ |
H2SO4 |
+ |
H2O |
CH3CH2OH |
+ |
H2SO4 |
+ |
[OH]– |
+ |
H2O |
+ |
+ |
NaOH(aq) |
CH3COONa |
+ |
CH3CH2OH |
+ |
H2O |
+ |
HCl |
+ |
H2O |
2 |
CH3CN |
+ |
2 |
H2O |
+ |
H+ |
CH3COOH |
+ |
[NH4]+ |
+ |
HCl(conc) |
+ |
H2O |
CH3COOH |
+ |
CH3OH |
[CoCl4]2– |
+ |
6 |
H2O |
[Co(H2O)6]2+ |
+ |
4 |
Cl– |
[VCl2(H2O)4]+ |
+ |
2 |
H2O |
[V(H2O)6]3+ |
+ |
2 |
Cl– |
+ |
2 |
H2O |
+ |
H2SO4(aq) |
+ |
NH3 |
[CoCl4]2– |
+ |
4 |
AgNO3 |
+ |
H2O |
4 |
AgCl |
+ |
Co2+ |
+ |
4 |
[NO3]– |
CH3COCl |
+ |
AgNO3 |
+ |
H2O |
AgCl |
+ |
CH3COOH |
+ |
H+ |
+ |
[NO3]– |
+ |
2 |
[OH]– |
+ |
2 |
Br– |
© Mark R. Leach 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com