
Interactions | Reactions | Processes
Classified as: Benzyne Mechanism
The benzyne mechanism is a type of strong base catalysed elimination-followed-by-addition aromatic substitution. A strong base, often NH2- in NH3, abstracts a proton which initiates the ejection of a nucleofuge so forming a highly reactive "benzyne", a type of highly strained alkyne. The benzyne reactive intermediate is highly susceptible to nucleophilic addition. Benzyne reaction products show characteristic substitution patterns.
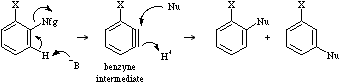
+ |
CO2 |
+ |
CO |
+ |
RLi |
+ |
LiF |
+ |
R–H |
+ |
N2 |
+ |
CO2 |
+ |
+ |
Li+ |
+ |
F– |
© Mark R. Leach 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com