
Interactions | Reactions | Processes
Classified as: Reduction Half Reaction
The electrochemical reduction of a species by Single Electron Transfer from an electrode can be assigned a voltage with respect to a standard hydrogen electrode.
During a reduction half reaction a species gains one or more electrons and these appear of the left hand side of the equation. By convention, redox half reactions are compared as the reduction half reaction. For more information look in the Chemogenesis webbook sections on redox chemistry and substitution, transfer, abstraction, displacement (STAD).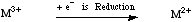
Al3+ |
+ |
3 |
e– |
Al |
Sb |
+ |
3 |
H+ |
+ |
3 |
e– |
H3Sb |
As |
+ |
3 |
H+ |
+ |
3 |
e– |
H3As |
Ba2+ |
+ |
2 |
e– |
Ba |
Be2+ |
+ |
2 |
e– |
Be |
2 |
HOBr |
+ |
2 |
H+ |
+ |
2 |
e– |
Br2 |
+ |
2 |
H2O |
Br2 |
+ |
2 |
e– |
2 |
Br– |
Cd2+ |
+ |
2 |
e– |
Cd |
Cs+ |
+ |
e– |
Cs |
Ca2+ |
+ |
2 |
e– |
Ca |
CO2 |
+ |
2 |
H+ |
+ |
2 |
e– |
CO |
+ |
H2O |
CO2 |
+ |
2 |
H+ |
+ |
2 |
e– |
HCOOH |
2 |
CO2 |
+ |
2 |
H+ |
+ |
2 |
e– |
Ce4+ |
+ |
e– |
Ce3+ |
Ce3+ |
+ |
3 |
e– |
Ce |
2 |
HClO |
+ |
2 |
H+ |
+ |
2 |
e– |
Cl2 |
+ |
2 |
H2O |
Cl2 |
+ |
2 |
e– |
2 |
Cl– |
[Cr2O7]2– |
+ |
14 |
H+ |
+ |
6 |
e– |
2 |
Cr3+ |
+ |
7 |
H2O |
Cr3+ |
+ |
e– |
Cr2+ |
Cr3+ |
+ |
3 |
e– |
Cr |
Cr2+ |
+ |
2 |
e– |
Cr |
Co3+ |
+ |
e– |
Co2+ |
Co2+ |
+ |
2 |
e– |
Co |
Cu2+ |
+ |
e– |
Cu+ |
Cu2+ |
+ |
Cl– |
+ |
e– |
CuCl |
Cu2+ |
+ |
2 |
e– |
Cu |
Cu+ |
+ |
e– |
Cu |
CuCl |
+ |
e– |
Cu |
+ |
Cl– |
F2 |
+ |
2 |
e– |
2 |
F– |
Ga3+ |
+ |
2 |
e– |
Ga+ |
Ga3+ |
+ |
3 |
e– |
Ga |
Ga2+ |
+ |
2 |
e– |
Ge |
Au3+ |
+ |
2 |
e– |
Au+ |
Au+ |
+ |
e– |
Au |
2 |
H+ |
+ |
2 |
e– |
H2 |
HOOH |
+ |
2 |
H+ |
+ |
2 |
e– |
2 |
H2O |
In3+ |
+ |
2 |
e– |
In+ |
In+ |
+ |
e– |
In |
2 |
[IO3]– |
+ |
12 |
H+ |
+ |
10 |
e– |
I2 |
+ |
6 |
H2O |
2 |
[IO]– |
+ |
4 |
H+ |
+ |
2 |
e– |
I2 |
+ |
2 |
H2O |
I2 |
+ |
2 |
e– |
+ |
H2O |
2 |
I– |
Fe3+ |
+ |
e– |
Fe2+ |
Fe3+ |
+ |
3 |
e– |
Fe |
Fe2+ |
+ |
2 |
e– |
Fe |
La3+ |
+ |
3 |
e– |
Pb4+ |
+ |
2 |
e– |
Pb2+ |
PbO2 |
+ |
4 |
H+ |
+ |
2 |
e– |
Pb2+ |
+ |
2 |
H2O |
PbO2 |
+ |
4 |
H+ |
Pb4+ |
+ |
2 |
H2O |
Pb2+ |
+ |
2 |
e– |
PbO |
+ |
2 |
H+ |
+ |
2 |
e– |
+ |
H2O |
PbSO4 |
+ |
2 |
e– |
+ |
[SO4]2– |
Li+ |
+ |
e– |
Li |
Mg2+ |
+ |
2 |
e– |
Mg |
[MnO4]– |
+ |
4 |
H+ |
+ |
3 |
e– |
MnO2 |
+ |
2 |
H2O |
[MnO4]– |
+ |
8 |
H+ |
+ |
5 |
e– |
Mg2+ |
+ |
4 |
H2O |
MnO2 |
+ |
4 |
H+ |
+ |
2 |
e– |
Mg2+ |
+ |
2 |
H2O |
Mg2+ |
+ |
2 |
e– |
Mn |
2 |
Hg2+ |
+ |
2 |
e– |
Hg23+ |
Hg2+ |
+ |
2 |
e– |
Hg |
Hg23+ |
+ |
2 |
e– |
2 |
Hg |
Hg2Cl2 |
+ |
2 |
e– |
2 |
Hg |
+ |
2 |
Cl– |
Ni2+ |
+ |
2 |
e– |
Ni |
2 |
[NO3]– |
+ |
4 |
H+ |
+ |
2 |
e– |
N2O4 |
+ |
2 |
H2O |
[NO3]– |
+ |
3 |
H+ |
+ |
2 |
e– |
HNO2 |
+ |
H2O |
[NO3]– |
+ |
4 |
H+ |
+ |
3 |
e– |
NO |
+ |
2 |
H2O |
[NO3]– |
+ |
10 |
H+ |
+ |
8 |
e– |
[NH4]+ |
+ |
3 |
H2O |
HNO2 |
+ |
H+ |
+ |
e– |
NO |
+ |
H2O |
2 |
NO2 |
+ |
8 |
H+ |
+ |
8 |
e– |
N2 |
+ |
4 |
H2O |
3 |
N2 |
+ |
2 |
H+ |
+ |
2 |
e– |
2 |
HN3 |
O2 |
+ |
4 |
H+ |
+ |
4 |
e– |
2 |
H2O |
O2 |
+ |
2 |
H+ |
+ |
2 |
e– |
HOOH |
O2 |
+ |
2 |
H2O |
+ |
4 |
e– |
4 |
[OH]– |
O3 |
+ |
2 |
H+ |
+ |
2 |
e– |
O2 |
+ |
H2O |
Pd2+ |
+ |
2 |
e– |
Pd |
H3PO4 |
+ |
2 |
H+ |
+ |
2 |
e– |
H3PO3 |
+ |
H2O |
H3PO3 |
+ |
2 |
H+ |
+ |
2 |
e– |
H3PO2 |
+ |
H2O |
4 |
H3PO2 |
+ |
4 |
H+ |
+ |
4 |
e– |
P |
+ |
8 |
H2O |
P |
+ |
12 |
H+ |
+ |
12 |
e– |
4 |
PH3 |
Pt2+ |
+ |
2 |
e– |
Pt |
K+ |
+ |
e– |
K |
Rb+ |
+ |
e– |
Rb |
Se |
+ |
2 |
H+ |
+ |
2 |
e– |
H2Se |
Se |
+ |
2 |
e– |
Se2– |
SiO2 |
+ |
4 |
H+ |
+ |
4 |
e– |
Si |
+ |
2 |
H2O |
Si |
+ |
4 |
H+ |
+ |
4 |
e– |
SiH4 |
Ag+ |
+ |
e– |
Ag |
Ag2O |
+ |
2 |
H+ |
+ |
2 |
e– |
2 |
Ag |
+ |
H2O |
AgCl |
+ |
e– |
Ag |
+ |
Cl– |
Na+ |
+ |
e– |
Na |
Sr2+ |
+ |
2 |
e– |
Sr |
[SO4]2– |
+ |
4 |
H+ |
+ |
2 |
e– |
H2SO3 |
+ |
H2O |
H2SO3 |
+ |
4 |
H+ |
+ |
4 |
e– |
S |
+ |
3 |
H2O |
S |
+ |
2 |
e– |
S2– |
S |
+ |
2 |
H+ |
+ |
2 |
e– |
H2S |
Te |
+ |
2 |
e– |
Te2– |
Tl3+ |
+ |
2 |
e– |
Tl+ |
Tl+ |
+ |
e– |
Tl |
Sn4+ |
+ |
2 |
e– |
Sn2+ |
Sn2+ |
+ |
2 |
e– |
Sn |
Ti3+ |
+ |
e– |
Ti2+ |
Ti2+ |
+ |
2 |
e– |
Ti |
U4+ |
+ |
e– |
U3+ |
U3+ |
+ |
3 |
e– |
U |
V3+ |
+ |
e– |
V2+ |
V2+ |
+ |
2 |
e– |
V |
Zn2+ |
+ |
2 |
e– |
Zn |
H2SO4 |
+ |
2 |
e– |
+ |
2 |
H+ |
SO2 |
+ |
2 |
H2O |
2 |
+ |
2 |
e– |
+ |
2 |
H+ |
[Cr(H2O)6]2+ |
+ |
e– |
[Cr(H2O)6]2+ |
[MnO4]– |
+ |
5 |
e– |
+ |
8 |
H+ |
+ |
2 |
H2O |
[Mn(H2O)6]2+ |
+ |
e– |
[ClO]– |
+ |
2 |
H+ |
+ |
2 |
e– |
Cl– |
+ |
H2O |
[NO3]– |
+ |
2 |
H+ |
+ |
e– |
NO2 |
+ |
H2O |
[SO4]2– |
+ |
4 |
H+ |
+ |
2 |
e– |
SO2 |
+ |
2 |
H2O |
O2 |
+ |
4 |
e– |
2 |
O2– |
H2SO4 |
+ |
8 |
H+ |
+ |
8 |
e– |
H2S |
+ |
4 |
H2O |
[SO4]2– |
+ |
10 |
H+ |
+ |
8 |
e– |
H2S |
+ |
4 |
H2O |
[ClO3]– |
+ |
6 |
H+ |
+ |
6 |
e– |
Cl– |
+ |
3 |
H2O |
+ |
2 |
e– |
2 |
[VO2]+ |
+ |
e– |
+ |
2 |
H+ |
+ |
4 |
H2O |
[VO]2+ |
[VO]2+ |
+ |
e– |
+ |
2 |
H+ |
V3+ |
+ |
6 |
H2O |
© Mark R. Leach 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com