
Interactions | Reactions | Processes
Classified as: Decomplexation or Fragmentation
Decomplexation or fragmentation is the reverse of complexation. However, where complexation is a bimolecular (second order) process, decomplexation is a unimolecular (first order) process.
Fragmentation into a Lewis acid/base pair is termed "heterolysis" and fragmentation into a pair of radicals is termed "homolysis". For more information look in the Chemogenesis webbook sections on fragmentation reactions.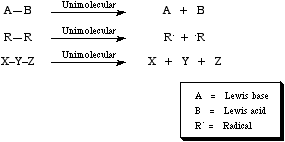
+ |
Pt |
+ |
H2 |
+ |
Pt |
+ |
3 |
H2 |
[H2O2]+• |
HO• |
+ |
[OH]– |
R–C–R |
+ |
+ |
+ |
+ |
+ |
+ |
+ |
+ |
(CH3)3CBr |
+ |
HBr |
+ |
(CH3)3N |
+ |
[CN]– |
+ |
[(CH3)3NH]+ |
H2C=CH2 |
+ |
RCOOH |
+ |
[OH]– |
+ |
(CH3)3N |
+ |
H2O |
(CH3)3C–NH2 |
+ |
HNO2 |
+ |
N2 |
+ |
H2O |
+ |
[OH]– |
R–NH2 |
+ |
HNO2 |
R+ |
+ |
N2 |
+ |
H2O |
+ |
[OH]– |
CH2N2 |
N2 |
+ |
CH2 |
+ |
HNO2 |
RCOOH |
+ |
N2 |
+ |
CO2 |
+ |
CO |
+ |
N2 |
+ |
CO2 |
2 |
H2NCl |
+ |
H2N–NH2 |
N2 |
+ |
2 |
NH4Cl |
3 |
H2NCl |
+ |
2 |
NH3 |
N2 |
+ |
3 |
NH4Cl |
4 |
H2N–OH |
+ |
H+ |
N2O |
+ |
2 |
NH3 |
+ |
3 |
H2O |
N2O3 |
+ |
2 |
H2SO4 |
2 |
[NO]+ |
+ |
2 |
[HSO4]– |
+ |
H2O |
2 |
HNO2 |
NO2 |
+ |
NO |
+ |
H2O |
H2C=C=O |
+ |
hν |
CH2 |
+ |
CO |
RCOO– |
R– |
+ |
CO2 |
R2C=C=O |
+ |
hν |
R–C–R |
+ |
CO |
2 |
R–C–R |
+ |
H2O |
+ |
HCl(aq) |
+ |
CO2 |
+ |
CH3CH2OH |
+ |
[SCN]– |
+ |
[NCO]– |
+ |
CO2 |
2 |
H2C=C=O |
NO2 |
+ |
hν |
NO |
+ |
O |
H2O |
+ |
hν |
H• |
+ |
HO• |
H2O |
+ |
hν |
H2 |
+ |
O |
HOOH |
+ |
hν |
2 |
HO• |
CO2 |
+ |
hν |
CO |
+ |
O |
H2C=CH2 |
+ |
hν |
HC≡CH |
+ |
H2 |
+ |
hν |
H2C=CH2 |
+ |
RCHO |
+ |
hν |
R–H |
+ |
CO |
+ |
hν |
2 |
H2C=CH2 |
+ |
CO |
+ |
hν |
+ |
CO |
O3 |
+ |
hν |
O |
+ |
O2 |
O3 |
+ |
O |
2 |
O2 |
+ |
Pt |
+ |
4 |
H2 |
2 |
H2O |
2 |
H2 |
+ |
O2 |
S2F10 |
SF6 |
+ |
SF4 |
SO2 |
+ |
H2O |
H2SO3 |
[Cr2O7]2– |
+ |
2 |
NaOH(aq) |
2 |
[CrO4]2– |
+ |
2 |
Na+ |
+ |
H2O |
[CH3CH2]+ |
+ |
H2O |
[CH3CH2]+ |
H2C=CH2 |
+ |
H+ |
[NO2]+ |
+ |
H2O |
+ |
H+ |
+ |
LiCH3 |
H2C=CH2 |
+ |
+ |
Li+ |
+ |
CH4 |
3 |
+ |
3 |
O2 |
CH3COONa |
+ |
NaOH |
CH4 |
+ |
Na2CO3 |
© Mark R. Leach 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com