
Interactions | Reactions | Processes
Classified as: Substitution: Nucleophilic Second Order
Second order nucleophilic substitution is a concerted processes in which a nucleophilic Lewis base displaces a nucleofugal Lewis base from a Lewis acid centre.
In organic chemistry, SN2 processes usually occur at a delta+ sp3 carbon centre. SN2 processes cause the tetrahedral carbon to invert like an umbrella via a five valent carbonium ion transition state. For more information look in the Chemogenesis webbook sections on substitution reactions and substitution, transfer, abstraction, displacement (STAD).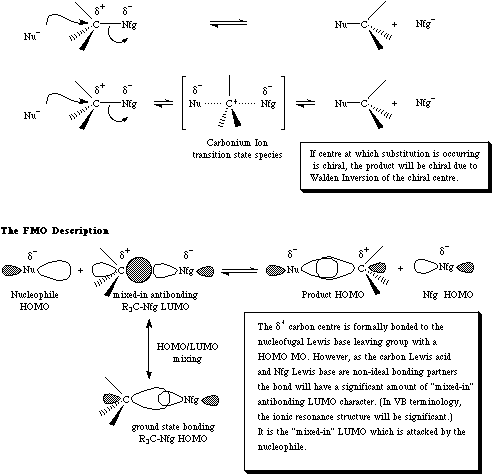
R–I |
+ |
RO– |
+ |
NaI |
RCH2CHO |
+ |
Br2 |
+ |
CH3COOH |
+ |
HBr |
RCOO– |
+ |
RCl |
+ |
Cl– |
RCOOH |
+ |
CH2N2 |
+ |
N2 |
RBr |
+ |
R–Nu |
+ |
Br– |
+ |
Br2 |
+ |
CH3COOH |
+ |
HBr |
R2CuLi |
+ |
RBr |
R–R |
+ |
RBr |
+ |
Br– |
R2S |
+ |
R–I |
[R3S]+ |
+ |
I– |
+ |
CH3I |
[R–S]– |
+ |
R–I |
R2S |
+ |
I– |
2 |
[R–S]– |
+ |
Br2 |
R–S–S–R |
+ |
2 |
Br– |
[R2OH]+ |
+ |
Br– |
RBr |
+ |
R–OH |
R–CH2–Br |
+ |
[(C6H5)3P–CH2R]+ |
+ |
Br– |
+ |
[OH]– |
+ |
H2S |
+ |
[OH]– |
+ |
NH3 |
+ |
RMgBr |
R–CH2CH2OH |
+ |
LiAlH4 |
+ |
2 |
[H] |
CH3CH2OH |
+ |
[OH]– |
+ |
[N3]– |
+ |
Cl– |
+ |
CH3COOH |
+ |
+ |
HCl |
CH3I |
+ |
[OH]– |
CH3OH |
+ |
I– |
CH3I |
+ |
(CH3)3N |
[(CH3)4N]+ |
+ |
I– |
[(CH3)4N]+ |
+ |
[OH]– |
CH3OH |
+ |
(CH3)3N |
[(CH3)4N]+ |
+ |
H2S |
[CH3–SH2]+ |
+ |
(CH3)3N |
(CH3)2CHBr |
+ |
CH3CH2O– |
+ |
CH3CH2OH |
CH3–CH=CH2 |
+ |
+ |
Br– |
+ |
CH3CH2OH |
+ |
[OH]– |
+ |
(CH3)3N |
CH2ClCH2Cl |
+ |
2 |
PhS(CH2)2SPh |
+ |
2 |
Cl– |
(CH3)2CHBr |
+ |
+ |
Br– |
+ |
CH3I |
+ |
I– |
+ |
Br2 |
+ |
[Et3O]+[BF4]– |
+ |
+ |
+ |
Li+ |
+ |
[BF4]– |
+ |
+ |
CH3CH2I |
+ |
+ |
Li+ |
+ |
I– |
CH3I |
+ |
[SCN]– |
N≡C–S–CH3 |
RCOCl |
+ |
[SCN]– |
CH3I |
+ |
[NO2]– |
CH3–NO2 |
CH3I |
+ |
+ |
R–I |
+ |
NaCN |
R–CN |
+ |
NaI |
R–I |
+ |
AgCN |
R–NC |
R2NH |
+ |
HNO2 |
R2N–NO |
[R–S]– |
+ |
N≡C–Cl |
R–S–C≡N |
+ |
Cl– |
2 |
R–SH |
+ |
N≡C–Br |
R–S–S–R |
+ |
HCN |
+ |
HBr |
RMgX |
+ |
N≡C–Cl |
R–CN |
+ |
Cl– |
RMgX |
+ |
I–C≡N |
R–I |
+ |
[CN]– |
+ |
[CN]– |
+ |
[R–S]– |
+ |
CH3COOH |
+ |
CH3CH2OH |
+ |
H+ |
+ |
H2O |
[BH4]– |
+ |
4 |
H2O |
4 |
H2 |
+ |
[B(OH)4]– |
+ |
H2O |
R–OH |
+ |
HCHO |
+ |
H+ |
+ |
H2O |
+ |
H+ |
+ |
H2O |
+ |
H+ |
+ |
H2O |
+ |
H+ |
+ |
H2O |
+ |
H+ |
+ |
H2O |
+ |
H+ |
+ |
H2O |
+ |
H+ |
R–C≡C– |
+ |
R–Nfg |
R–C≡C–R |
+ |
H– |
RCHO |
+ |
+ |
Cl– |
RCOCl |
+ |
+ |
+ |
+ |
RCOO– |
(RCOO)2O |
+ |
+ |
+ |
+ |
[H2N]– |
+ |
+ |
[OH]– |
RCOOH |
+ |
+ |
[CH3–O]– |
+ |
+ |
R– |
+ |
+ |
H2O |
RCOOH |
+ |
H+ |
+ |
+ |
R–Nfg |
+ |
Li+ |
+ |
R–Nfg |
+ |
[SCN]– |
R–S–C≡N |
+ |
R–N=C=S |
+ |
R–TeH |
+ |
R–Nfg |
H2Te |
+ |
RMgX |
+ |
SO2Cl2 |
RSO2Cl |
RSO2Cl |
+ |
H2O |
R–SO3H |
+ |
HCl |
[R–SO2]– |
+ |
RCl |
+ |
Cl– |
[R–Se]– |
+ |
R–Nfg |
R2Sb |
+ |
[R3O]+ |
+ |
R–Nu |
+ |
+ |
I– |
+ |
Li+ |
+ |
RCOO– |
+ |
Li+ |
+ |
CH3I |
+ |
R–Nfg |
+ |
R–Nfg |
+ |
[NCO]– |
R–N=C=O |
+ |
RO– |
+ |
N≡C–Cl |
R–O–C≡N |
+ |
Cl– |
R+ |
+ |
[NCO]– |
R–N=C=O |
R–NH2 |
+ |
R–N=C=O |
+ |
2 |
HCl |
+ |
Br2 |
+ |
CH3COOH |
+ |
HBr |
+ |
(CH3CO)2CO |
+ |
CH3COOH |
+ |
(CH3CO)2CO |
+ |
CH3COOH |
Cl2O |
+ |
H2O |
2 |
HClO |
SiCl4 |
+ |
4 |
CH3MgI |
Si(CH3)4 |
+ |
4 |
Mg2+ |
+ |
4 |
Cl– |
+ |
4 |
I– |
N≡C–C≡N |
+ |
2 |
[OH]– |
[NCO]– |
+ |
[CN]– |
+ |
H2O |
H2NCl |
+ |
NH3 |
+ |
NaOH |
H2N–NH2 |
+ |
NaCl |
+ |
H2O |
2 |
H2NCl |
+ |
H2N–NH2 |
N2 |
+ |
2 |
NH4Cl |
3 |
H2NCl |
+ |
2 |
NH3 |
N2 |
+ |
3 |
NH4Cl |
PCl3 |
+ |
3 |
+ |
3 |
Cl– |
PCl3 |
+ |
3 |
+ |
3 |
Cl– |
PCl3 |
+ |
3 |
[(CH3)3C]– |
+ |
3 |
Cl– |
PCl3 |
+ |
3 |
R– |
R3P |
+ |
3 |
Cl– |
PCl3 |
+ |
3 |
[CH3]– |
(CH3)3P |
+ |
3 |
Cl– |
PCl3 |
+ |
3 |
[CH3CH2]– |
(CH3CH2)3P |
+ |
3 |
Cl– |
Cl2 |
+ |
[OH]– |
[ClO]– |
+ |
Cl– |
+ |
H+ |
Br2 |
+ |
[OH]– |
[BrO]– |
+ |
Br– |
+ |
H+ |
I2 |
+ |
[OH]– |
[IO]– |
+ |
I– |
+ |
H+ |
+ |
NaOH |
+ |
H2O |
+ |
NaCl |
+ |
H2O |
+ |
+ |
2 |
Na |
+ |
CH3CH2OH |
+ |
2 |
NaBr |
+ |
H2 |
+ |
NaOH |
+ |
H2O |
+ |
NaCl |
+ |
H2O |
+ |
[SCN]– |
+ |
CH3CH2OH |
+ |
H2O |
+ |
[NCO]– |
+ |
[SCN]– |
+ |
[NCO]– |
+ |
CO2 |
+ |
LiI |
+ |
CH2Cl2 |
+ |
.880 NH3 |
+ |
H2O |
+ |
(NH4)2CO3 |
+ |
CO2 |
+ |
2 |
H2O |
+ |
NH3 |
2 |
+ |
Ca(OH)2 |
2 |
+ |
CaCl2 |
+ |
2 |
H2O |
1000 |
+ |
1000 |
Polycarbonate |
+ |
2000 |
HCl |
+ |
+ |
HCl |
+ |
CH3NH2 |
+ |
HCl |
+ |
H2 |
+ |
2 |
NaOH |
+ |
2 |
NaCl |
CH2ClCH2Cl |
+ |
NH3 |
+ |
CaO |
+ |
H2O |
+ |
CaCl2 |
CH2ClCH2Cl |
+ |
4 |
NH3 |
+ |
2 |
NH4Cl |
+ |
NH3 |
+ |
H2O |
+ |
HCN |
+ |
[OH]– |
+ |
NaOH |
+ |
NaCl |
+ |
H2O |
+ |
NaOH |
+ |
NaCl |
+ |
HNO3|H2SO4 concentrated |
+ |
H2 |
+ |
1000 |
+ |
1000 |
Terylene |
+ |
1000 |
H2O |
CH3CH2Br |
+ |
2 |
NH3 |
CH3CH2NH2 |
+ |
NH4Br |
CH3Br |
+ |
KCN |
CH3CN |
+ |
KBr |
+ |
I2 |
+ |
HI |
+ |
2 |
NH3 |
+ |
NH4Cl |
CH2ClCH2Cl |
+ |
2 |
KCN |
+ |
2 |
KCl |
+ |
2 |
KCN |
+ |
2 |
KBr |
CH3NH2 |
+ |
3 |
CH3CH2Br |
+ |
Br– |
+ |
2 |
HBr |
+ |
[OH]– |
+ |
HBr |
+ |
KCN |
+ |
KBr |
CH3–CH=CH2 |
+ |
Br2 |
+ |
Br– |
(CH3)2CHBr |
+ |
2 |
NH3 |
(CH3)2CHNH2 |
+ |
NH4Br |
CH3CH2OH |
+ |
2 |
(CH3)2CHBr |
+ |
NaOH(aq) |
+ |
NaBr |
2 |
+ |
2 |
NaOH |
+ |
CH3–CH=CH2 |
+ |
2 |
NaCl |
+ |
H2O |
CH3CH2OH |
+ |
Polyethylene Glycol |
CH3Br |
+ |
[CN]– |
CH3CN |
+ |
Br– |
CH2ClCH2Cl |
+ |
2 |
[CN]– |
+ |
2 |
Cl– |
+ |
2 |
[CN]– |
+ |
2 |
Br– |
(CH3)2CHBr |
+ |
KOH |
+ |
H2O |
+ |
KBr |
2 |
(CH3)2CHBr |
+ |
2 |
KOH |
+ |
CH3–CH=CH2 |
+ |
2 |
KBr |
+ |
H2O |
+ |
[CN]– |
+ |
+ |
2 |
NH3 |
+ |
NH4Br |
+ |
CH3CH2OH |
+ |
[CN]– |
+ |
Cl– |
+ |
2 |
[OH]– |
+ |
2 |
Br– |
© Mark R. Leach 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com