
Interactions | Reactions | Processes
Classified as: Radical Addition
Radical addition involves a radical, Ri, interacting with pi-system to form a sigma bond and a new radical centre.
The newly formed radical centre then abstracts an atom X from X-R to give the addition product and another molecule of Ri. Radical additions propagate in this cyclic or "chain" manner until reagents are consumed or the reaction is terminated by radical coupling. Radical additions require a catalytic quantity of "initiator" (a substance which easily fragments into radicals, such as dibenzoyl peroxide) plus heat or UV light to get the reaction going. For more information look in the Chemogenesis webbook section on addition reactions.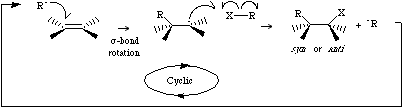
+ |
HBr |
+ |
Br• |
CH3–CH=CH2 |
+ |
HBr |
+ |
R• |
2 |
PCl3 |
+ |
O2 |
2 |
POCl3 |
1000 |
CH3–CH=CH2 |
+ |
Ziegler-Natta catalyst |
1000 |
F2C=CF2 |
1000 |
H2C=CHCl |
1000 |
1000 |
1000 |
1000 |
1000 |
1000 |
+ |
Ziegler-Natta catalyst |
1000 |
–[CH2CH2]n– |
+ |
+ |
(C6H5COO)2 |
H2C=CH2 |
+ |
HBr |
+ |
γ |
CH3CH2Br |
1000 |
+ |
1000 |
Styrene-Butadiene Rubber |
1000 |
Polybutadiene Rubber |
1000 |
+ |
1000 |
Nitrile Rubber |
1000 |
+ |
1000 |
+ |
1000 |
ABS |
2 |
HC≡CH |
+ |
HCl(aq) |
+ |
CuCl |
+ |
H2O |
1000 |
H2C=CCl2 |
1000 |
+ |
H2O |
+ |
(C6H5COO)2 |
1000 |
H2C=CH2 |
–[CH2CH2]n– |
© Mark R. Leach 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com