
Interactions | Reactions | Processes
Classified as: Addition: Electrophilic Protonation
The addition of Brønsted is a stepwise process. The first step involves a electrophilic proton complexing with a pi-system to generate Type 4 Lewis complex which collapses to a carbenium ion centre. This subsequently complexes with a nucleophilic Lewis base, usually, but not always, the counter ion of the Brønsted acid used to initiate the reaction.
For more information look in the Chemogenesis webbook section on addition reactions.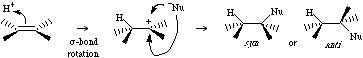
+ |
HCl |
+ |
HBr |
+ |
HI |
R–C≡C–R |
+ |
HCl |
+ |
HCl |
+ |
CO |
+ |
H2O |
+ |
H+ |
+ |
H+(aq) |
(CH3)3C–COOH |
RCHO |
+ |
R–OH |
+ |
+ |
H2O |
+ |
R–OH |
+ |
+ |
H2O |
+ |
2 |
R–OH |
+ |
+ |
2 |
H2O |
H+ |
+ |
O=C=C=C=O |
+ |
2 |
HCl |
+ |
HF |
+ |
HCl |
+ |
HBr |
(CH3)3CBr |
+ |
HI |
(CH3)3CI |
+ |
H2O |
+ |
H2SO4 |
(CH3)3COH |
CH3–CH=CH2 |
+ |
HF |
CH3–CH=CH2 |
+ |
HCl |
CH3–CH=CH2 |
+ |
HBr |
(CH3)2CHBr |
CH3–CH=CH2 |
+ |
HI |
(CH3)2CHI |
CH3–CH=CH2 |
+ |
H2O |
+ |
H+ |
H2C=CH2 |
+ |
HF |
H2C=CH2 |
+ |
HCl |
+ |
AlCl3 |
CH3CH2Cl |
H2C=CH2 |
+ |
HBr |
+ |
AlBr3 |
CH3CH2Br |
H2C=CH2 |
+ |
HI |
CH3CH2I |
H2C=CH2 |
+ |
H2O |
+ |
H+ |
CH3CH2OH |
HC≡CH |
+ |
H2O |
+ |
H+ |
CH3CHO |
HC≡CH |
+ |
HCl |
+ |
HgCl2 |
H2C=CHCl |
H2C=CH2 |
+ |
HClO |
CH3–CH=CH2 |
+ |
HClO |
+ |
CH3OH |
+ |
HCN |
+ |
[OH]– |
+ |
CH3OH |
+ |
H2SO4 |
+ |
H2O |
+ |
[NH4]+ |
+ |
[HSO4]– |
+ |
H2O |
+ |
2 |
HCN |
+ |
HCl |
+ |
CO |
+ |
H2O |
+ |
H+ |
HC≡CH |
+ |
H2O |
+ |
H+ |
CH3CHO |
2 |
H2C=CH2 |
+ |
H2SO4 |
+ |
Ag2SO4 |
(C2H5O)2SO2 |
CH3COOH |
+ |
H2C=CH2 |
+ |
+ |
+ |
3 |
H2O |
+ |
H2SO4 |
3 |
H2C=CHCl |
+ |
HCl |
+ |
FeCl3 |
CH3CHCl2 |
CH3CHCl2 |
+ |
Cl2 |
+ |
SiO2 |
CH3CCl3 |
+ |
HCl |
+ |
H2O |
+ |
H+ |
CH3CHO |
+ |
HCN |
+ |
H2O |
+ |
H2O |
+ |
H+ |
2 |
+ |
2 |
HCN |
+ |
2 |
H2 |
+ |
Pd on BaSO4 |
+ |
2 |
H2O |
+ |
2 |
+ |
2 |
HCN |
+ |
2 |
H2 |
+ |
Pd on BaSO4 |
+ |
2 |
H2O |
+ |
2 |
+ |
2 |
HCN |
+ |
2 |
H2 |
+ |
Pd on BaSO4 |
+ |
2 |
H2O |
+ |
2 |
+ |
2 |
HCN |
+ |
2 |
H2 |
+ |
Pd on BaSO4 |
+ |
2 |
H2O |
+ |
2 |
+ |
2 |
HCN |
+ |
2 |
H2 |
+ |
Pd on BaSO4 |
+ |
2 |
H2O |
+ |
2 |
+ |
2 |
HCN |
+ |
2 |
H2 |
+ |
Pd on BaSO4 |
+ |
2 |
H2O |
+ |
2 |
+ |
2 |
HCN |
+ |
2 |
H2 |
+ |
Pd on BaSO4 |
+ |
2 |
H2O |
+ |
+ |
HBr |
+ |
H2SO4 |
H2C=CH2 |
+ |
H2SO4 |
+ |
HBr |
© Mark R. Leach 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com