
Interactions | Reactions | Processes
Classified as: Ethylene the Petrochemical Building Block
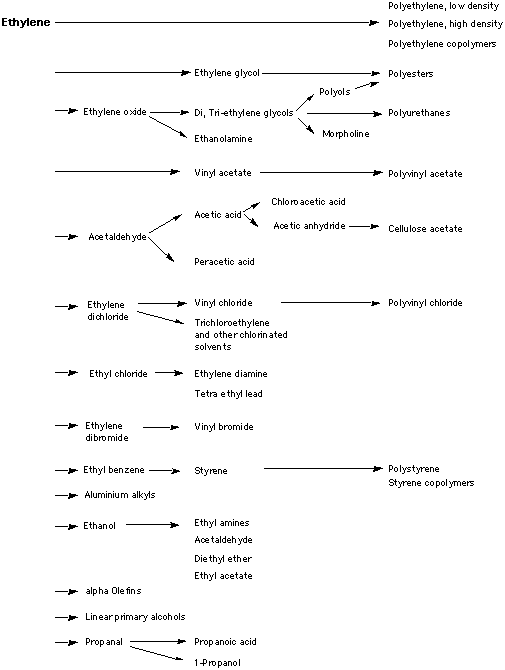
Ethylene, produced as a side reaction while cracking long chain alkanes, is the main starting material for the petrochemical industry. A large petrochemical plant may produce several thousand products, all starting from ethylene.
+ |
CH3CH2Cl |
+ |
AlCl3 |
+ |
H+ |
+ |
[AlCl4]– |
2 |
H2C=CH2 |
+ |
SCl2 |
S(CH2CH2Cl)2 |
1000 |
H2C=CH2 |
+ |
Ziegler-Natta catalyst |
–[CH2CH2]n– |
CH3CH3 |
H2C=CH2 |
+ |
H2 |
Al(CH2CH3)3 |
+ |
TiCl4 |
Ziegler-Natta catalyst |
H2C=CH2 |
+ |
HCl |
+ |
AlCl3 |
CH3CH2Cl |
H2C=CH2 |
+ |
HBr |
+ |
AlBr3 |
CH3CH2Br |
H2C=CH2 |
+ |
H2O |
+ |
H+ |
CH3CH2OH |
+ |
FeO.Fe2O3 |
+ |
H2 |
2 |
H2C=CH2 |
+ |
O2 |
+ |
H2O |
+ |
PdCl2 |
+ |
CuCl2 |
2 |
CH3CHO |
H2C=CH2 |
+ |
PdCl2 |
+ |
H2O |
CH3CHO |
+ |
Pd |
+ |
2 |
HCl |
H2C=CH2 |
+ |
HClO |
2 |
+ |
Na2CO3 |
+ |
H2O |
2 |
+ |
CO2 |
+ |
2 |
NaCl |
2 |
+ |
Ca(OH)2 |
2 |
+ |
CaCl2 |
+ |
2 |
H2O |
2 |
H2C=CH2 |
+ |
O2 |
+ |
Ag |
2 |
4 |
H2C=CH2 |
+ |
2 |
O2 |
+ |
6 |
CH3COOH |
2 |
+ |
2 |
+ |
2 |
H2O |
2 |
+ |
2 |
+ |
6 |
H2O |
4 |
+ |
6 |
CH3COOH |
2 |
H2C=CH2 |
+ |
O2 |
+ |
2 |
CH3COOH |
+ |
PdCl2 |
2 |
+ |
2 |
H2O |
H2C=CH2 |
+ |
Cl2 |
+ |
FeCl3 |
CH2ClCH2Cl |
2 |
H2C=CH2 |
+ |
4 |
HCl |
+ |
O2 |
+ |
CuCl2 |
2 |
CH2ClCH2Cl |
+ |
2 |
H2O |
CH2ClCH2Cl |
H2C=CHCl |
+ |
HCl |
H2C=CH2 |
+ |
Br2 |
+ |
HBr |
H2C=CH2 |
+ |
HBr |
+ |
γ |
CH3CH2Br |
2 |
Al |
+ |
3 |
Mg |
+ |
6 |
CH3CH2Cl |
2 |
Al(CH2CH3)3 |
+ |
3 |
MgCl2 |
AlH3 |
+ |
3 |
H2C=CH2 |
Al(CH2CH3)3 |
H2C=CH2 |
+ |
CO |
+ |
H2 |
2 |
+ |
O2 |
2 |
CH3CH2COOH |
+ |
H2 |
+ |
Pd on C |
1000 |
+ |
1000 |
Terylene |
+ |
2000 |
H2O |
1000 |
H2C=CH2 |
–[CH2CH2]n– |
© Mark R. Leach 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com