
Interactions | Reactions | Processes
Classified as: Change of Coordination Number
Centres can expand or contract their coordination numbers.
For more information look in the Chemogenesis webbook section on the different types of reaction chemistry.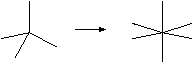
SiF4 |
+ |
2 |
F– |
[SiF6]2– |
GeF4 |
+ |
2 |
F– |
[GeF6]2– |
GeCl4 |
+ |
2 |
Cl– |
[GeCl6]2– |
SnF4 |
+ |
2 |
F– |
[SnF6]2– |
SnCl4 |
+ |
2 |
Cl– |
[SnCl6]2– |
PbF4 |
+ |
2 |
F– |
[PbF6]2– |
PbCl4 |
+ |
2 |
Cl– |
[PbCl6]2– |
PCl3 |
+ |
Cl2 |
PCl5 |
5 |
PF3 |
+ |
5 |
Br2 |
3 |
PF5 |
+ |
2 |
PBr5 |
PBr3 |
+ |
Br2 |
PBr5 |
SbCl3 |
+ |
Cl2 |
SbCl5 |
UF4 |
+ |
F2 |
UF6 |
UF4 |
+ |
ClF3 |
UF6 |
+ |
ClF |
Rn |
+ |
F2 |
RnF2 |
Kr |
+ |
F2 |
KrF2 |
Xe |
+ |
F2 |
XeF2 |
Xe |
+ |
2 |
F2 |
XeF4 |
Xe |
+ |
3 |
F2 |
XeF6 |
XeF2 |
+ |
H2 |
Xe |
+ |
2 |
HF |
XeF4 |
+ |
2 |
H2 |
Xe |
+ |
4 |
HF |
XeF6 |
+ |
3 |
H2 |
Xe |
+ |
6 |
HF |
2 |
SF4 |
+ |
XeF4 |
2 |
SF6 |
+ |
Xe |
Pt |
+ |
3 |
XeF2 |
+ |
HF |
PtF6 |
+ |
3 |
Xe |
CH3I |
+ |
XeF2 |
CH3IF |
+ |
Xe |
2 |
XeF2 |
+ |
2 |
H2O |
2 |
Xe |
+ |
4 |
HF |
+ |
O2 |
6 |
XeF4 |
+ |
12 |
H2O |
2 |
XeO3 |
+ |
4 |
Xe |
+ |
24 |
HF |
+ |
3 |
O2 |
RbF |
+ |
XeF6 |
Rb+ |
+ |
[XeF7]– |
CsF |
+ |
XeF6 |
Cs+ |
+ |
[XeF7]– |
Ar |
+ |
HF |
+ |
hν |
H–Ar–F |
2 |
FeCl3 |
2 |
FeCl2 |
+ |
Cl2 |
[Cu(H2O)6]2+ |
+ |
4 |
Cl– |
[CuCl4]2– |
+ |
6 |
H2O |
[Co(H2O)6]2+ |
+ |
4 |
Cl– |
[CoCl4]2– |
+ |
6 |
H2O |
[MnO4]– |
+ |
5 |
e– |
+ |
8 |
H+ |
+ |
2 |
H2O |
[Mn(H2O)6]2+ |
[Al(H2O)6]3+ |
+ |
4 |
NaOH(aq) |
[Al(OH)4]– |
+ |
4 |
Na+ |
+ |
6 |
H2O |
[Co(H2O)6]2+ |
+ |
4 |
HCl(conc) |
[CoCl4]2– |
+ |
4 |
H+ |
+ |
6 |
H2O |
PCl5 |
PCl3 |
+ |
Cl2 |
© Mark R. Leach 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com