
Interactions | Reactions | Processes
Classified as: Ligand Exchange (Inorganic)
A useful rule, devised by Linus Pauling, is that the most stable arrangement of ligands about central atom is for the atom with highest electronegativity to have the lowest electronegativity ligands, and vice versa. This relationship is seen in many ligand exchange reactions.
For more information look in the Chemogenesis webbook sections on electronegativity, substitution reactions and substitution, transfer, abstraction, displacement (STAD).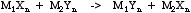
BBr3 |
+ |
PCl3 |
BCl3 |
+ |
PBr3 |
2 |
HI |
+ |
Cl2 |
2 |
HCl |
+ |
I2 |
B(OCH3)3 |
+ |
4 |
NaH |
NaBH4 |
+ |
3 |
NaOCH3 |
[BH4]– |
+ |
H+ |
+ |
3 |
H2O |
H3BO3 |
+ |
4 |
H2 |
AlCl3 |
+ |
4 |
LiH |
LiAlH4 |
+ |
3 |
LiCl |
SiCl4 |
+ |
LiAlH4 |
SiH4 |
+ |
LiCl |
+ |
AlCl3 |
B2O3 |
+ |
2 |
Al |
+ |
3 |
H2 |
+ |
AlCl3 |
B2H6 |
+ |
Al2O3 |
4 |
+ |
3 |
NaBH4 |
+ |
2 |
B2H6 |
+ |
3 |
NaBF4 |
+ |
4 |
2 |
NaOH |
+ |
H2S |
Na2S |
+ |
2 |
H2O |
3 |
Na2SO3 |
+ |
2 |
POCl3 |
3 |
SOCl2 |
+ |
6 |
Na+ |
+ |
2 |
[PO4]3– |
Ca2+ |
+ |
2 |
[OH]– |
+ |
CO2 |
+ |
H2O |
CaCO3 |
+ |
H2O |
CaCO3 |
+ |
CO2 |
+ |
H2O |
+ |
H2O |
Ca2+ |
+ |
2 |
[HCO3]– |
3 |
[SO3]2– |
+ |
2 |
POCl3 |
3 |
SOCl2 |
+ |
2 |
[PO4]3– |
LiOH |
+ |
H2S |
LiSH |
+ |
H2O |
NaOH |
+ |
H2S |
NaSH |
+ |
H2O |
KOH |
+ |
H2S |
KSH |
+ |
H2O |
RbOH |
+ |
H2S |
RbSH |
+ |
H2O |
CsOH |
+ |
H2S |
CsSH |
+ |
H2O |
2 |
LiOH |
+ |
H2S |
Li2S |
+ |
2 |
H2O |
2 |
KOH |
+ |
H2S |
K2S |
+ |
2 |
H2O |
2 |
RbOH |
+ |
H2S |
Rb2S |
+ |
2 |
H2O |
2 |
CsOH |
+ |
H2S |
Cs2S |
+ |
2 |
H2O |
Li2S |
+ |
H2O |
LiSH |
+ |
LiOH |
Na2S |
+ |
H2O |
NaSH |
+ |
NaOH |
K2S |
+ |
H2O |
KSH |
+ |
KOH |
Rb2S |
+ |
H2O |
RbSH |
+ |
RbOH |
Cs2S |
+ |
H2O |
CsSH |
+ |
CsOH |
2 |
LiNH2 |
+ |
NH3 |
LiNH2 |
+ |
NH3 |
MgO |
+ |
3 |
C |
MgC2 |
+ |
CO |
SrO |
+ |
3 |
C |
SrC2 |
+ |
CO |
BaO |
+ |
3 |
C |
BaC2 |
+ |
CO |
Al(OH)3 |
+ |
3 |
NaOH |
+ |
6 |
HF |
Na3AlF6 |
+ |
6 |
H2O |
B2O3 |
+ |
3 |
CaF2 |
+ |
3 |
H2SO4 |
2 |
BF3 |
+ |
3 |
CaSO4 |
+ |
3 |
H2O |
B2O3 |
+ |
6 |
CH3OH |
2 |
B(OCH3)3 |
+ |
3 |
H2O |
H3BO3 |
+ |
2 |
[NH4]+ [FHF]– |
NH4BF4 |
+ |
3 |
H2O |
+ |
NH3 |
6 |
NH4BF4 |
+ |
B2O3 |
8 |
BF3 |
+ |
3 |
H2O |
+ |
6 |
NH3 |
H3BO3 |
+ |
4 |
HF |
+ |
H2O |
H+ [BF4]– |
+ |
3 |
H2O |
H3BO3 |
+ |
4 |
HF |
+ |
H2O |
H+ |
+ |
[BF4]– |
+ |
3 |
H2O |
B2H6 |
+ |
2 |
NH3 |
[H3N–BH2–NH3]+ [BH4]– |
B2H6 |
+ |
6 |
CH3OH |
2 |
B(OCH3)3 |
+ |
6 |
H2 |
B2H6 |
+ |
6 |
Cl2 |
2 |
BCl3 |
+ |
6 |
HCl |
B2H6 |
+ |
6 |
Br2 |
2 |
BBr3 |
+ |
6 |
HBr |
BF3 |
+ |
3 |
CH3MgI |
(CH3)3B |
+ |
3 |
Mg2+ |
+ |
3 |
F– |
+ |
3 |
I– |
CH4 |
+ |
4 |
S |
CS2 |
+ |
2 |
H2S |
SiCl4 |
+ |
4 |
LiH |
SiH4 |
+ |
4 |
LiCl |
GeCl4 |
+ |
LiAlH4 |
+ |
GeH4 |
+ |
LiCl |
+ |
AlCl3 |
GeO2 |
+ |
NaBH4 |
+ |
H2O |
GeH4 |
+ |
NaBO2 Na3[B3O6] |
SnCl4 |
+ |
LiAlH4 |
+ |
SnH4 |
+ |
LiCl |
+ |
AlCl3 |
SiO2 |
+ |
6 |
HF |
2 |
H+ |
+ |
[SiF6]2– |
+ |
2 |
H2O |
PbO2 |
+ |
4 |
CH3COOH |
Pb(OCOCH3)4 |
+ |
2 |
H2O |
CO2 |
+ |
SiF4 |
CF4 |
+ |
SiO2 |
CS2 |
+ |
3 |
Cl2 |
+ |
FeCl3 |
CCl4 |
+ |
Cl–S–S–Cl |
CS2 |
+ |
2 |
Cl–S–S–Cl |
+ |
FeCl3 |
CCl4 |
+ |
6 |
S |
3 |
CS2 |
+ |
6 |
Cl2 |
+ |
FeCl3 |
3 |
CCl4 |
+ |
6 |
S |
CCl2F2 |
+ |
F2 |
CF4 |
+ |
Cl2 |
CCl4 |
+ |
2 |
HF |
+ |
SbF3 |
+ |
SbCl5 |
CCl2F2 |
+ |
2 |
HCl |
CCl4 |
+ |
H2O |
+ |
2 |
HCl |
2 |
Ca3(PO4)2 |
+ |
6 |
SiO2 |
2 |
P2O5 |
+ |
6 |
CaSiO3 |
PCl3 |
+ |
3 |
H2O |
H3PO3 |
+ |
3 |
HCl |
POCl3 |
+ |
3 |
CH3CH2OH |
(CH3CH2O)3P=O |
+ |
3 |
HCl |
3 |
RCOOH |
+ |
PCl3 |
3 |
RCOCl |
+ |
H3PO3 |
3 |
R–OH |
+ |
PCl3 |
3 |
RCl |
+ |
H3PO3 |
NH4Cl |
+ |
3 |
Cl2 |
NCl3 |
+ |
4 |
HCl |
7 |
.880 NH3 |
+ |
3 |
I2 |
NI3.6NH3 |
+ |
3 |
HI |
PCl3 |
+ |
3 |
HF |
PF3 |
+ |
3 |
HCl |
PCl3 |
+ |
AsF3 |
PF3 |
+ |
AsCl3 |
P2O5 |
+ |
5 |
CaF2 |
2 |
PF5 |
+ |
5 |
CaO |
3 |
PCl5 |
+ |
5 |
AsF3 |
3 |
PF5 |
+ |
5 |
AsCl3 |
2 |
As2O3 |
+ |
10 |
F2 |
4 |
AsF5 |
+ |
3 |
O2 |
Sb2O3 |
+ |
6 |
HF |
+ |
H2O |
2 |
SbF3 |
+ |
3 |
H2O |
2 |
Sb2O3 |
+ |
10 |
F2 |
4 |
SbF5 |
+ |
3 |
O2 |
SbCl5 |
+ |
5 |
HF |
SbF5 |
+ |
5 |
HCl |
PCl5 |
+ |
4 |
H2O |
H3PO4 |
+ |
5 |
HCl |
PCl5 |
+ |
H2O |
POCl3 |
+ |
2 |
HCl |
ZnCl2 |
+ |
2 |
N2O4 |
Zn(NO3)2 |
+ |
2 |
NOCl |
NO2 |
+ |
CO |
NO |
+ |
CO2 |
N2O4 |
+ |
2 |
CO |
2 |
NO |
+ |
2 |
CO2 |
N2O5 |
+ |
NaCl |
Cl–NO2 |
+ |
NaNO3 |
5 |
H3PO4 |
+ |
POCl3 |
3 |
+ |
3 |
HCl |
Na2CO3 |
+ |
SO2 |
+ |
H2O |
Na2SO3 |
+ |
CO2 |
SOCl2 |
+ |
2 |
HBr |
SOBr2 |
+ |
2 |
HCl |
3 |
SOCl2 |
+ |
2 |
SbF3 |
3 |
SOF2 |
+ |
2 |
SbCl3 |
Ag2CO3 |
+ |
2 |
HF |
2 |
AgF |
+ |
CO2 |
+ |
H2O |
2 |
Na2O2 |
+ |
2 |
CO2 |
2 |
Na2CO3 |
+ |
O2 |
TeF6 |
+ |
6 |
H2O |
Te(OH)6 |
+ |
6 |
HF |
Te(OH)6 |
+ |
6 |
HI |
TeI4 |
+ |
I2 |
+ |
6 |
H2O |
SeCl4 |
+ |
4 |
AgF |
SeF4 |
+ |
4 |
AgCl |
4 |
BCl3 |
+ |
3 |
SF4 |
4 |
BF3 |
+ |
3 |
Cl2 |
+ |
3 |
SCl2 |
SF4 |
+ |
2 |
H2O |
SO2 |
+ |
4 |
HF |
SCl4 |
+ |
2 |
H2O |
SO2 |
+ |
4 |
HCl |
6 |
SCl2 |
+ |
16 |
NH3 |
S4N4 |
+ |
12 |
NH4Cl |
+ |
2 |
S |
UO2 |
+ |
4 |
HF |
UF4 |
+ |
2 |
H2O |
Al2O3 |
+ |
6 |
HF |
2 |
AlF3 |
+ |
3 |
H2O |
2 |
I2O5 |
+ |
10 |
F2 |
4 |
IF5 |
+ |
5 |
O2 |
3 |
UO2 |
+ |
4 |
BrF3 |
3 |
UF4 |
+ |
2 |
Br2 |
+ |
3 |
O2 |
SiO2 |
+ |
2 |
BrF5 |
SiF4 |
+ |
2 |
BrF3 |
+ |
O2 |
CH3OH |
+ |
HCl |
CH3Cl |
+ |
H2O |
Fe3O4 |
+ |
8 |
HCl |
FeCl2 |
+ |
2 |
FeCl3 |
+ |
4 |
H2O |
(NH4)2S2O8 |
+ |
2 |
NaOH |
Na2S2O8 |
+ |
2 |
NH3 |
+ |
2 |
H2O |
CaO |
+ |
SiO2 |
CaSiO3 |
2 |
CaF2 |
+ |
SiO2 |
+ |
2 |
H2SO4 |
SiF4 |
+ |
2 |
CaSO4 |
+ |
2 |
H2O |
3 |
SiF4 |
+ |
2 |
H2O |
2 |
H2SiF6(aq) |
+ |
SiO2 |
Al2O3 |
+ |
6 |
HF |
2 |
AlF3 |
+ |
3 |
H2O |
6 |
NH4F |
+ |
3 |
NaOH |
+ |
Al(OH)3 |
Na3AlF6 |
+ |
6 |
NH3 |
+ |
6 |
H2O |
Na2B4O7 |
+ |
6 |
CaF2 |
+ |
7 |
SO3 |
+ |
H2SO4 |
4 |
BF3 |
+ |
6 |
CaSO4 |
+ |
Na2SO4 |
H3BO3 |
+ |
3 |
HF |
+ |
H2SO4 |
BF3 |
+ |
3 |
H2O |
H3BO3 |
+ |
3 |
HSO3F |
BF3 |
+ |
3 |
H2SO4 |
H2S |
+ |
ZnO |
ZnS |
+ |
H2O |
[Cu(H2O)6]2+ |
+ |
[Cu(EDTA)]2– |
+ |
6 |
H2O |
[Cr(H2O)6]2+ |
+ |
6 |
NH3(aq) |
[Cr(NH3)6]2+ |
+ |
6 |
H2O |
[Cu(H2O)6]2+ |
+ |
4 |
NH3(aq) |
[Cu(NH3)4(H2O)2]2+ |
+ |
4 |
H2O |
[Cu(H2O)6]2+ |
+ |
4 |
Cl– |
[CuCl4]2– |
+ |
6 |
H2O |
[Co(H2O)6]2+ |
+ |
4 |
Cl– |
[CoCl4]2– |
+ |
6 |
H2O |
[MnO4]– |
+ |
5 |
e– |
+ |
8 |
H+ |
+ |
2 |
H2O |
[Mn(H2O)6]2+ |
[Fe(H2O)6]2+ |
+ |
3 |
+ |
6 |
H2O |
+ |
+ |
6 |
+ |
3 |
+ |
6 |
[Fe(H2O)6]3+ |
+ |
3 |
[C2O4]2– |
[Fe(C2O4)3]3– |
+ |
6 |
H2O |
[Fe(H2O)6]3+ |
+ |
[SCN]– |
[Fe(H2O)5(SCN)]2+ |
+ |
H2O |
[CrCl2(H2O)4]+ |
+ |
Zn |
+ |
2 |
H2O |
[Cr(H2O)6]2+ |
+ |
Zn2+ |
+ |
2 |
Cl– |
[Co(H2O)6]2+ |
+ |
6 |
NH3(aq) |
[Co(NH3)6]2+ |
+ |
6 |
H2O |
[Cu(H2O)6]2+ |
+ |
4 |
NH3(aq) |
[Cu(NH3)4(H2O)2]2+ |
+ |
4 |
H2O |
[Cr(H2O)6]2+ |
+ |
6 |
NaOH(aq) |
[Cr(OH)6]3– |
+ |
6 |
H2O |
+ |
6 |
Na+ |
Cr(H2O)3(OH)3 |
+ |
6 |
NH3(aq) |
[Cr(NH3)6]2+ |
+ |
3 |
H2O |
+ |
3 |
[OH]– |
[Co(H2O)6]2+ |
+ |
4 |
HCl(conc) |
[CoCl4]2– |
+ |
4 |
H+ |
+ |
6 |
H2O |
AgCl |
+ |
2 |
NH3(aq) |
[Ag(NH3)2]+ |
+ |
Cl– |
[Ag(NH3)2]+ |
+ |
NaBr |
+ |
H2O |
AgBr |
+ |
2 |
NH3 |
+ |
Na+ |
AgBr |
+ |
2 |
Na2S2O3 |
+ |
H2O |
[Ag(S2O3)2]3– |
+ |
4 |
Na+ |
+ |
Br– |
AgBr |
+ |
2 |
KCN |
+ |
H2O |
[Ag(CN)2]– |
+ |
2 |
K+ |
+ |
Br– |
CaCO3 |
+ |
SiO2 |
CaSiO3 |
+ |
CO2 |
© Mark R. Leach 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com