
Interactions | Reactions | Processes
Classified as: Substitution: Electrophilic Second Order
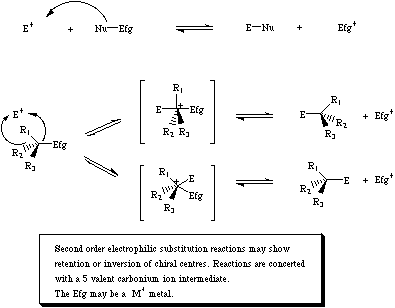
Second order electrophilic substitution involves a Lewis base transferring from one Lewis acid (Efg) to another (E).
RCl |
+ |
AlCl3 |
R+ [AlCl4]– |
RCOCl |
+ |
AlCl3 |
+ |
Br2 |
+ |
CH3COOH |
+ |
HBr |
+ |
CO |
+ |
HCl |
+ |
AlCl3 |
+ |
[AlCl4]– |
+ |
H+ |
+ |
Br2 |
+ |
LiBr |
+ |
ClCOOCH3 |
+ |
LiCl |
H3BO3 |
+ |
H2O |
[B(OH)4]– |
+ |
H+ |
Al(OH)3 |
+ |
H2O |
[Al(OH)4]– |
+ |
H+ |
RCOCl |
+ |
AlCl3 |
[RCO]+ |
+ |
[AlCl4]– |
+ |
AlCl3 |
+ |
[AlCl4]– |
SbF5 |
+ |
HSO3F |
ROH |
+ |
SO3 |
BF3 |
+ |
ClF3 |
[ClF2]+ |
+ |
[BF4]– |
SbF5 |
+ |
ClF3 |
[ClF2]+ |
+ |
[SbF6]– |
BrF5 |
+ |
CsF |
Cs+ |
+ |
[BrF6]– |
IF5 |
+ |
KF |
K+ |
+ |
[IF6]– |
IF7 |
+ |
CsF |
Cs+ |
+ |
[IF8]– |
I–Cl |
+ |
KCl |
K+ |
+ |
[ICl2]– |
I–Cl |
+ |
KI |
K+ |
+ |
[I2Cl]– |
XeF2 |
+ |
HF |
[XeF]+ |
+ |
[FHF]– |
XeF2 |
+ |
AsF5 |
[XeF]+ |
+ |
[AsF6]– |
XeF2 |
+ |
PF5 |
[XeF]+ |
+ |
[PF6]– |
XeF2 |
+ |
SbF5 |
[XeF]+ |
+ |
[SbF6]– |
RbF |
+ |
XeF6 |
Rb+ |
+ |
[XeF7]– |
CsF |
+ |
XeF6 |
Cs+ |
+ |
[XeF7]– |
ZrC |
+ |
3 |
Pt |
ZrPt3 |
+ |
C |
+ |
H2C=CH2 |
+ |
HCl |
+ |
AlCl3 |
+ |
+ |
HCl |
+ |
AlCl3 |
+ |
AlCl3 |
+ |
[AlCl4]– |
+ |
+ |
AlCl3 |
+ |
HCl |
© Mark R. Leach 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com