
Interactions | Reactions | Processes
Classified as: Substitution: Electrophilic Aromatic
Electrophilic aromatic substitution involves an electrophilic Lewis acid interacting with an aromatic pi-system Lewis base to form a Wheland intermediate.
This high energy species then rapidly ejects an electrofugal Lewis acid (usually H+) to form the substituted aromatic. This analysis is explained in detail in the Chemogenesis webbook.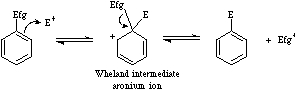
+ |
Cl+ |
+ |
H+ |
+ |
Cl+ / [AlCl4]– |
+ |
H+ |
+ |
[AlCl4]– |
+ |
Br+ |
+ |
H+ |
+ |
Br+ [FeBr4]– |
+ |
H+ |
+ |
[FeBr4]– |
+ |
I2 |
+ |
CuCl2 |
+ |
[NO2]+ |
+ |
H+ |
+ |
HNO3 |
+ |
H2SO4 |
+ |
H2O |
+ |
H2S2O7 |
+ |
H2SO4 |
+ |
H2S2O7 |
+ |
H2O |
+ |
H2SO4 |
+ |
HSO3Cl |
+ |
H2O |
+ |
R+ |
+ |
H+ |
+ |
RCl |
+ |
AlCl3 |
+ |
[AlCl4]– |
+ |
[RCO]+ |
+ |
RCOCl |
+ |
AlCl3 |
+ |
H+ |
+ |
[AlCl4]– |
+ |
Cl2 |
+ |
AlCl3 |
+ |
H+ |
+ |
[AlCl4]– |
+ |
+ |
+ |
[AlCl4]– |
+ |
H+ |
CO2 |
+ |
+ |
+ |
BF3 |
+ |
H+ |
+ |
[BF4]– |
+ |
CH3CH2Cl |
+ |
AlCl3 |
+ |
H+ |
+ |
[AlCl4]– |
+ |
+ |
H+ |
+ |
SOCl2 |
+ |
AlCl3 |
+ |
CS2 |
+ |
+ |
H2O |
+ |
PCl5 |
+ |
SnCl4 |
+ |
+ |
+ |
AlCl3 |
+ |
H2O |
+ |
CH3–CH=CH2 |
2 |
+ |
+ |
H2SO4 |
+ |
H2O |
+ |
3 |
HNO3 |
+ |
3 |
H2O |
+ |
HNO3|H2SO4 concentrated |
+ |
H2O |
+ |
+ |
AlCl3 |
+ |
H2O |
2 |
+ |
2 |
CO |
+ |
FeCl3 |
+ |
CO |
+ |
FeCl3 |
2 |
+ |
CO |
+ |
FeCl3 |
+ |
+ |
AlCl3 |
+ |
HCl |
+ |
H2C=CH2 |
+ |
HCl |
+ |
AlCl3 |
2 |
+ |
CCl3CHO |
+ |
H+ |
DDT |
+ |
H2O |
+ |
NH3 |
+ |
NiO |
+ |
Ni |
+ |
H2 |
+ |
HNO3|H2SO4 concentrated |
+ |
H2 |
+ |
+ |
CH3COCl |
+ |
AlCl3 |
+ |
HCl |
+ |
[CH3CO]+ |
+ |
H+ |
+ |
HNO3 |
+ |
H2SO4 |
+ |
H2O |
+ |
+ |
AlCl3 |
+ |
HCl |
© Mark R. Leach 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com