
Interactions | Reactions | Processes
Classified as: Oxidation: Of Substrate By Reagent
A substrate is deemed to be oxidised by an oxidising agent if the oxidising agent (1) removes electrons by single electron transfer or SET, or (2) removes electron rich hydrogen from the substrate, or (3) removes electron density from the substrate by reversing polarisation by adding electronegative oxygen or (4) electronegative halogen atoms.
For more information look in the Chemogenesis webbook section on redox chemistry.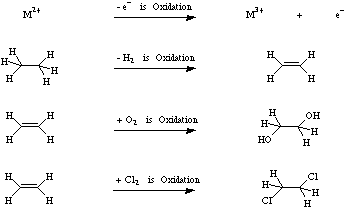
RCH2OH |
+ |
RCHO |
RCH2OH |
+ |
CrO3 |
RCOOH |
+ |
RCHO |
+ |
CrO3 |
RCOOH |
RCH2CHO |
+ |
Br2 |
+ |
CH3COOH |
+ |
HBr |
CH4 |
+ |
Cl2 |
+ |
hν |
CH3Cl |
+ |
HCl |
+ |
Cl2 |
+ |
Br2 |
+ |
+ |
R–C≡C–H |
+ |
HgSO4 |
+ |
H+(aq) |
R–C≡C–H |
+ |
+ |
HOOH |
+ |
NaOH |
RCH2CHO |
+ |
KMnO4 |
+ |
KMnO4 |
+ |
OH–(aq) |
+ |
RCOOH |
+ |
(KSO3)2NO |
R2S |
+ |
+ |
2 |
[R–S]– |
+ |
Br2 |
R–S–S–R |
+ |
2 |
Br– |
2 |
NO |
+ |
O2 |
2 |
NO2 |
CH4 |
+ |
2 |
O2 |
CO2 |
+ |
2 |
H2O |
2 |
+ |
13 |
O2 |
8 |
CO2 |
+ |
10 |
H2O |
+ |
Fe3+ |
+ |
Fe2+ |
R3N |
+ |
HOOH |
R3N=O |
R2S |
+ |
HOOH |
R2S |
+ |
2 |
HOOH |
+ |
2 |
H2O |
+ |
HOOH |
+ |
HOOH |
H2N–NH2 |
+ |
2 |
HOOH |
N2 |
+ |
4 |
H2O(g) |
+ |
6.5 |
O2 |
4 |
CO2 |
+ |
5 |
H2O(g) |
Cu |
+ |
4 |
HNO3 5 molar |
Cu(NO3)2 |
+ |
2 |
NO2 |
+ |
2 |
H2O |
Sn |
+ |
4 |
HNO3 |
Sn2O |
+ |
4 |
NO2 |
+ |
2 |
H2O |
2 |
Mg |
+ |
O2 |
2 |
MgO |
4 |
Al |
+ |
3 |
O2 |
2 |
Al2O3 |
C |
+ |
O2 |
CO2 |
S |
+ |
O2 |
SO2 |
+ |
10 |
Se |
+ |
10 |
H2Se |
RCOOH |
+ |
HOOH |
+ |
H2O |
CH3COOH |
+ |
HOOH |
+ |
H2SO4 |
CH3COOOH |
+ |
H2O |
2 |
NaBH4 |
+ |
I2 |
+ |
B2H6 |
+ |
H2 |
+ |
2 |
NaI |
2 |
NaOH |
+ |
2 |
F2 |
+ |
H2O |
OF2 |
+ |
2 |
NaF |
+ |
H2O |
2 |
Na |
+ |
2 |
H2O |
2 |
NaOH |
+ |
H2 |
2 |
K |
+ |
2 |
H2O |
2 |
KOH |
+ |
H2 |
6 |
Li |
+ |
N2 |
2 |
Li3N |
Li |
+ |
NH3 |
Li+ |
+ |
e–(am) |
Na |
+ |
NH3 |
Na+ |
+ |
e–(am) |
K |
+ |
NH3 |
K+ |
+ |
e–(am) |
4 |
Li |
+ |
O2 |
2 |
Li2O |
2 |
Na |
+ |
O2 |
Na2O2 |
K |
+ |
O2 |
K2O |
CO |
+ |
Na2O2 |
Na2CO3 |
4 |
Li |
+ |
O2 |
+ |
NH3 |
2 |
Li2O |
2 |
Li |
+ |
O2 |
+ |
NH3 |
Li2O2 |
Li |
+ |
O2 |
+ |
NH3 |
LiO2 |
4 |
Na |
+ |
O2 |
+ |
NH3 |
2 |
Na2O |
2 |
Na |
+ |
O2 |
+ |
NH3 |
Na2O2 |
Na |
+ |
O2 |
+ |
NH3 |
NaO2 |
4 |
K |
+ |
O2 |
+ |
NH3 |
2 |
KO2 |
2 |
K |
+ |
O2 |
+ |
NH3 |
K2O2 |
K |
+ |
O2 |
+ |
NH3 |
K2O |
4 |
Rb |
+ |
O2 |
+ |
NH3 |
2 |
Rb2O |
2 |
Rb |
+ |
O2 |
+ |
NH3 |
Rb2O2 |
Rb |
+ |
O2 |
+ |
NH3 |
RbO2 |
4 |
Cs |
+ |
O2 |
+ |
NH3 |
2 |
Cs2O |
2 |
Cs |
+ |
O2 |
+ |
NH3 |
Cs2O2 |
Cs |
+ |
O2 |
+ |
NH3 |
CsO2 |
2 |
Na2S |
+ |
2 |
O2 |
+ |
H2O |
Na2S2O3 |
+ |
2 |
NaOH |
2 |
Li |
+ |
2 |
C |
LiC≡CLi |
4 |
HCl |
+ |
MnO2 |
Cl2 |
+ |
MnCl2 |
+ |
2 |
H2O |
4 |
HCl |
+ |
O2 |
+ |
CuCl2 |
2 |
Cl2 |
+ |
2 |
H2O |
2 |
Li |
+ |
H2 |
2 |
LiH |
2 |
Na |
+ |
H2 |
2 |
NaH |
2 |
K |
+ |
H2 |
2 |
KH |
2 |
Rb |
+ |
H2 |
2 |
RbH |
2 |
Cs |
+ |
H2 |
2 |
CsH |
Be |
+ |
H2 |
BeH2 |
Mg |
+ |
H2 |
MgH2 |
Ca |
+ |
H2 |
CaH2 |
Sr |
+ |
H2 |
SrH2 |
Ba |
+ |
H2 |
BaH2 |
2 |
Li |
+ |
2 |
H2O |
2 |
LiOH |
+ |
H2 |
2 |
Rb |
+ |
2 |
H2O |
2 |
RbOH |
+ |
H2 |
2 |
Cs |
+ |
2 |
H2O |
2 |
CsOH |
+ |
H2 |
Mg |
+ |
H2O |
MgO |
+ |
H2 |
Ca |
+ |
2 |
H2O |
Ca(OH)2 |
+ |
H2 |
Sr |
+ |
2 |
H2O |
Sr(OH)2 |
+ |
H2 |
Ba |
+ |
2 |
H2O |
Ba(OH)2 |
+ |
H2 |
2 |
Li |
+ |
2 |
H+ |
2 |
Li+ |
+ |
H2 |
2 |
Na |
+ |
2 |
H+ |
2 |
Na+ |
+ |
H2 |
2 |
K |
+ |
2 |
H+ |
2 |
K+ |
+ |
H2 |
2 |
Rb |
+ |
2 |
H+ |
2 |
Rb+ |
+ |
H2 |
2 |
Cs |
+ |
2 |
H+ |
2 |
Cs+ |
+ |
H2 |
Be |
+ |
2 |
H+ |
Be2+ |
+ |
H2 |
Mg |
+ |
2 |
H+ |
Mg2+ |
+ |
H2 |
Ca |
+ |
2 |
H+ |
Ca2+ |
+ |
H2 |
Sr |
+ |
2 |
H+ |
Sr2+ |
+ |
H2 |
Ba |
+ |
2 |
H+ |
Ba2+ |
+ |
H2 |
2 |
Be |
+ |
O2 |
2 |
BeO |
2 |
Ca |
+ |
O2 |
2 |
CaO |
2 |
Sr |
+ |
O2 |
2 |
SrO |
2 |
Ba |
+ |
O2 |
2 |
BaO |
Ba |
+ |
O2 |
BaO2 |
2 |
BaO |
+ |
O2 |
2 |
BaO2 |
Be |
+ |
2 |
NaOH |
+ |
2 |
H2O |
2 |
Na+ |
+ |
[Be(OH)4]2– |
+ |
H2 |
12 |
Li |
+ |
P |
4 |
Li3P |
3 |
Li |
+ |
As |
Li3As |
3 |
Li |
+ |
Sb |
Li3Sb |
12 |
Na |
+ |
P |
4 |
Na3P |
3 |
Na |
+ |
As |
Na3As |
3 |
Na |
+ |
Sb |
Na3Sb |
12 |
K |
+ |
P |
4 |
K3P |
3 |
K |
+ |
As |
K3As |
3 |
K |
+ |
Sb |
K3Sb |
12 |
Rb |
+ |
P |
4 |
Rb3P |
3 |
Rb |
+ |
As |
Rb3As |
3 |
Rb |
+ |
Sb |
Rb3Sb |
12 |
Cs |
+ |
P |
4 |
Cs3P |
3 |
Cs |
+ |
As |
Cs3As |
3 |
Cs |
+ |
Sb |
Cs3Sb |
2 |
Li |
+ |
F2 |
2 |
LiF |
2 |
Li |
+ |
Cl2 |
2 |
LiCl |
2 |
Li |
+ |
Br2 |
2 |
LiBr |
2 |
Li |
+ |
I2 |
2 |
LiI |
2 |
Na |
+ |
F2 |
2 |
NaF |
2 |
Na |
+ |
Cl2 |
2 |
NaCl |
2 |
Na |
+ |
Br2 |
2 |
NaBr |
2 |
Na |
+ |
I2 |
2 |
NaI |
2 |
K |
+ |
F2 |
2 |
KF |
2 |
K |
+ |
Cl2 |
2 |
KCl |
2 |
K |
+ |
Br2 |
2 |
KBr |
2 |
K |
+ |
I2 |
2 |
KI |
2 |
Rb |
+ |
F2 |
2 |
RbF |
2 |
Rb |
+ |
Cl2 |
2 |
RbCl |
2 |
Rb |
+ |
Br2 |
2 |
RbBr |
2 |
Rb |
+ |
I2 |
2 |
RbI |
2 |
Cs |
+ |
F2 |
2 |
CsF |
2 |
Cs |
+ |
Cl2 |
2 |
CsCl |
2 |
Cs |
+ |
Br2 |
2 |
CsBr |
2 |
Cs |
+ |
I2 |
2 |
CsI |
10 |
Li |
+ |
15 |
S |
10 |
Li+ |
+ |
S2– |
+ |
–S–S– |
+ |
[S–S–S]2– |
+ |
[S–S–S–S]2– |
+ |
[S–S–S–S–S]2– |
10 |
Na |
+ |
15 |
S |
10 |
Na+ |
+ |
S2– |
+ |
–S–S– |
+ |
[S–S–S]2– |
+ |
[S–S–S–S]2– |
+ |
[S–S–S–S–S]2– |
10 |
K |
+ |
15 |
S |
10 |
K+ |
+ |
S2– |
+ |
–S–S– |
+ |
[S–S–S]2– |
+ |
[S–S–S–S]2– |
+ |
[S–S–S–S–S]2– |
10 |
Rb |
+ |
15 |
S |
10 |
Rb+ |
+ |
S2– |
+ |
–S–S– |
+ |
[S–S–S]2– |
+ |
[S–S–S–S]2– |
10 |
Cs |
+ |
15 |
S |
10 |
Cs+ |
+ |
S2– |
+ |
–S–S– |
+ |
[S–S–S]2– |
+ |
[S–S–S–S]2– |
+ |
[S–S–S–S–S]2– |
2 |
Li |
+ |
Se |
Li2Se |
2 |
Na |
+ |
Se |
Na2Se |
2 |
K |
+ |
Se |
K2Se |
2 |
Rb |
+ |
Se |
Rb2Se |
2 |
Cs |
+ |
Se |
Cs2Se |
2 |
Li |
+ |
Te |
Li2Te |
2 |
Na |
+ |
Te |
Na2Te |
2 |
K |
+ |
Te |
K2Te |
2 |
Rb |
+ |
Te |
Rb2Te |
2 |
Cs |
+ |
Te |
Cs2Te |
Rb |
+ |
NH3 |
Rb+ |
+ |
e–(am) |
Cs |
+ |
NH3 |
Cs+ |
+ |
e–(am) |
2 |
Li |
+ |
2 |
NH3 |
+ |
Fe2+ |
+ |
NH3 |
2 |
LiNH2 |
+ |
H2 |
2 |
Na |
+ |
2 |
NH3 |
+ |
Fe2+ |
+ |
NH3 |
2 |
NaNH2 |
+ |
H2 |
2 |
K |
+ |
2 |
NH3 |
+ |
Fe2+ |
+ |
NH3 |
2 |
KNH2 |
+ |
H2 |
2 |
Rb |
+ |
2 |
NH3 |
+ |
Fe2+ |
+ |
NH3 |
2 |
RbNH2 |
+ |
H2 |
2 |
Cs |
+ |
2 |
NH3 |
+ |
Fe2+ |
+ |
NH3 |
2 |
CsNH2 |
+ |
H2 |
2 |
Al |
+ |
3 |
Na2O2 |
Al2O3 |
+ |
3 |
Na2O |
3 |
Be |
+ |
N2 |
Be3N2 |
3 |
Mg |
+ |
N2 |
Mg3N2 |
3 |
Ca |
+ |
N2 |
Ca3N2 |
3 |
Sr |
+ |
N2 |
Sr3N2 |
3 |
Ba |
+ |
N2 |
Ba3N2 |
6 |
Be |
+ |
P |
2 |
Be3P2 |
6 |
Mg |
+ |
P |
2 |
Mg3P2 |
6 |
Ca |
+ |
P |
2 |
Ca3P2 |
6 |
Sr |
+ |
P |
2 |
Sr3P2 |
6 |
Ba |
+ |
P |
2 |
Ba3P2 |
Be |
+ |
S |
BeS |
Mg |
+ |
S |
MgS |
Ca |
+ |
S |
CaS |
Sr |
+ |
S |
SrS |
Ba |
+ |
S |
BaS |
Be |
+ |
Se |
BeS |
Mg |
+ |
Se |
MgSe |
Ca |
+ |
Se |
CaSe |
Sr |
+ |
Se |
SrSe |
Ba |
+ |
Se |
BaSe |
Be |
+ |
Te |
BeTe |
Mg |
+ |
Te |
MgTe |
Ca |
+ |
Te |
CaTe |
Sr |
+ |
Te |
SrTe |
Ba |
+ |
Te |
BaTe |
Be |
+ |
F2 |
BeF2 |
Be |
+ |
Cl2 |
BeCl2 |
Be |
+ |
Br2 |
BeBr2 |
Be |
+ |
I2 |
BeI2 |
Mg |
+ |
F2 |
MgF2 |
Mg |
+ |
Cl2 |
MgCl2 |
Mg |
+ |
Br2 |
MgBr2 |
Mg |
+ |
I2 |
MgI2 |
Ca |
+ |
F2 |
CaF2 |
Ca |
+ |
Cl2 |
CaCl2 |
Ca |
+ |
Br2 |
CaBr2 |
Ca |
+ |
I2 |
CaI2 |
Sr |
+ |
F2 |
SrF2 |
Sr |
+ |
Cl2 |
SrCl2 |
Sr |
+ |
Br2 |
SrBr2 |
Sr |
+ |
I2 |
SrI2 |
Ba |
+ |
F2 |
BaF2 |
Ba |
+ |
Cl2 |
BaCl2 |
Ba |
+ |
Br2 |
BaBr2 |
Ba |
+ |
I2 |
BaI2 |
Be |
+ |
2 |
NH3 |
Be(NH2)2 |
+ |
H2 |
Mg |
+ |
2 |
NH3 |
Mg(NH2)2 |
+ |
H2 |
Ca |
+ |
2 |
NH3 |
Ca(NH2)2 |
+ |
H2 |
Sr |
+ |
2 |
NH3 |
Sr(NH2)2 |
+ |
H2 |
Ba |
+ |
2 |
NH3 |
Ba(NH2)2 |
+ |
H2 |
Mg |
+ |
Air |
MgO |
+ |
Mg3N2 |
Ca |
+ |
2 |
C |
CaC2 |
Mg |
+ |
2 |
C |
MgC2 |
Sr |
+ |
2 |
C |
SrC2 |
Ba |
+ |
2 |
C |
BaC2 |
4 |
B |
+ |
3 |
O2 |
2 |
B2O3 |
2 |
B |
+ |
3 |
S |
B2S3 |
2 |
B |
+ |
2 |
NH3 |
2 |
BNx Hexagonal |
+ |
3 |
H2 |
2 |
B |
+ |
3 |
F2 |
2 |
BF3 |
2 |
B |
+ |
3 |
Cl2 |
2 |
BCl3 |
2 |
B |
+ |
3 |
Br2 |
2 |
BBr3 |
2 |
B |
+ |
3 |
I2 |
2 |
BI3 |
2 |
B |
+ |
6 |
NaOH |
2 |
Na3BO3 |
+ |
3 |
H2 |
4 |
Ga |
+ |
3 |
O2 |
2 |
Ga2O3 |
4 |
In |
+ |
3 |
O2 |
2 |
In2O3 |
4 |
Tl |
+ |
2 |
O2 |
Tl2O3 |
+ |
Tl2O |
2 |
Al |
+ |
N2 |
2 |
AlN |
2 |
Al |
+ |
3 |
F2 |
2 |
AlF3 |
2 |
Ga |
+ |
3 |
F2 |
2 |
GaF3 |
2 |
In |
+ |
3 |
F2 |
2 |
InF3 |
2 |
Al |
+ |
3 |
Cl2 |
2 |
AlCl3 |
2 |
Ga |
+ |
3 |
Cl2 |
2 |
GaCl3 |
2 |
In |
+ |
3 |
Cl2 |
2 |
InCl3 |
2 |
Tl |
+ |
2 |
Cl2 |
TlCl3 |
+ |
TlCl |
2 |
Tl |
+ |
2 |
Br2 |
TlBr3 |
+ |
TlBr |
2 |
Al |
+ |
3 |
Br2 |
2 |
AlBr3 |
2 |
Ga |
+ |
3 |
Br2 |
2 |
GaBr3 |
2 |
In |
+ |
3 |
Br2 |
2 |
InBr3 |
2 |
Tl |
+ |
3 |
I2 |
2 |
TlI3 |
2 |
B |
+ |
3 |
I2 |
2 |
BI3 |
2 |
Al |
+ |
3 |
I2 |
2 |
AlI3 |
2 |
Ga |
+ |
3 |
I2 |
2 |
GaI3 |
2 |
In |
+ |
3 |
I2 |
2 |
InI3 |
2 |
In |
+ |
3 |
H2 |
2 |
InH3 |
2 |
Ga |
+ |
3 |
H2 |
2 |
GaH3 |
2 |
Al |
+ |
6 |
H+ |
2 |
Al3+ |
+ |
3 |
H2 |
2 |
Ga |
+ |
6 |
H+ |
2 |
Ga3+ |
+ |
3 |
H2 |
2 |
In |
+ |
6 |
H+ |
2 |
In3+ |
+ |
3 |
H2 |
2 |
Tl |
+ |
6 |
H+ |
2 |
Tl3+ |
+ |
3 |
H2 |
2 |
Al |
+ |
6 |
H2O |
+ |
2 |
[OH]– |
2 |
[Al(OH)4]– |
+ |
3 |
H2 |
2 |
Al |
+ |
2 |
NaOH |
+ |
6 |
H2O |
2 |
Na[Al(OH)4] |
+ |
3 |
H2 |
2 |
NaBO2 Na3[B3O6] |
+ |
2 |
HOOH |
+ |
6 |
H2O |
Na2 [B2(OH)4(O2)2] .6H2O |
B2H6 |
+ |
6 |
Cl2 |
2 |
BCl3 |
+ |
6 |
HCl |
B2H6 |
+ |
6 |
Br2 |
2 |
BBr3 |
+ |
6 |
HBr |
Si |
+ |
2 |
Cl2 |
SiCl4 |
5 |
CO |
+ |
I2O5 |
5 |
CO2 |
+ |
I2 |
2 |
C |
+ |
Air |
2 |
CO + 2N2 producer gas |
Fe |
+ |
5 |
CO |
Fe(CO)5 |
CO |
+ |
S |
O=C=S |
+ |
6 |
O2 |
+ |
Yeast |
6 |
CO2 |
+ |
6 |
H2O |
CH4 |
+ |
4 |
S |
CS2 |
+ |
2 |
H2S |
SiO2 |
+ |
2 |
F2 |
SiF4 |
+ |
O2 |
2 |
Mg |
+ |
Si |
Mg2Si |
Ge |
+ |
2 |
Cl2 |
GeCl4 |
NaNH2 |
+ |
C |
NaCN |
+ |
H2 |
CH4 |
+ |
NH3 |
+ |
Pt on carbon |
HCN |
+ |
3 |
H2 |
2 |
CH4 |
+ |
2 |
NH3 |
+ |
3 |
O2 |
+ |
Pt on carbon |
+ |
Rh on Carbon |
2 |
HCN |
+ |
6 |
H2O |
4 |
Ag |
+ |
8 |
NaCN |
+ |
O2 |
+ |
2 |
H2O |
4 |
Na[Ag(CN)2] |
+ |
4 |
NaOH |
4 |
Au |
+ |
8 |
NaCN |
+ |
O2 |
+ |
2 |
H2O |
4 |
Na[Au(CN)2] |
+ |
4 |
NaOH |
4 |
[CN]– |
+ |
2 |
Cu2+ |
N≡C–C≡N |
+ |
2 |
CuCN |
SiC |
+ |
4 |
F2 |
CF4 |
+ |
SiF4 |
CS2 |
+ |
3 |
Cl2 |
+ |
FeCl3 |
CCl4 |
+ |
Cl–S–S–Cl |
CS2 |
+ |
2 |
Cl–S–S–Cl |
+ |
FeCl3 |
CCl4 |
+ |
6 |
S |
3 |
CS2 |
+ |
6 |
Cl2 |
+ |
FeCl3 |
3 |
CCl4 |
+ |
6 |
S |
CCl2F2 |
+ |
F2 |
CF4 |
+ |
Cl2 |
8 |
NH3 |
+ |
3 |
Br2 |
+ |
H2O |
N2 |
+ |
6 |
NH4Br |
P |
+ |
5 |
O2 |
2 |
P2O5 |
4 |
NH3 |
+ |
3 |
O2 |
2 |
N2 |
+ |
6 |
H2O |
PH3 |
+ |
2 |
O2 |
H3PO4 |
H2N–NH2 |
+ |
O2 |
N2 |
+ |
2 |
H2O |
NH3 |
+ |
NaOCl(aq) |
H2NCl |
+ |
NaOH |
PCl3 |
+ |
Cl2 |
PCl5 |
2 |
PCl3 |
+ |
O2 |
2 |
POCl3 |
3 |
PCl3 |
+ |
P2O5 |
+ |
3 |
Cl2 |
5 |
POCl3 |
NH4Cl |
+ |
3 |
Cl2 |
NCl3 |
+ |
4 |
HCl |
7 |
.880 NH3 |
+ |
3 |
I2 |
NI3.6NH3 |
+ |
3 |
HI |
P |
+ |
6 |
Cl2 |
4 |
PCl3 |
P |
+ |
6 |
Br2 |
4 |
PBr3 |
2 |
As |
+ |
5 |
F2 |
2 |
AsF5 |
2 |
As2O3 |
+ |
10 |
F2 |
4 |
AsF5 |
+ |
3 |
O2 |
2 |
Sb2O3 |
+ |
10 |
F2 |
4 |
SbF5 |
+ |
3 |
O2 |
2 |
Sb |
+ |
3 |
Cl2 |
2 |
SbCl3 |
2 |
Bi |
+ |
5 |
F2 |
2 |
BiF5 |
3 |
PCl5 |
+ |
P2O5 |
5 |
POCl3 |
3 |
Cu |
+ |
8 |
HNO3(aq) |
3 |
Cu(NO3)2 |
+ |
2 |
NO |
+ |
4 |
H2O |
2 |
NO |
+ |
Cl2 |
2 |
NOCl |
4 |
NO |
+ |
O2 |
2 |
N2O3 |
2 |
NO2 |
+ |
Cl2 |
2 |
Cl–NO2 |
N2O4 |
+ |
Cl2 |
2 |
Cl–NO2 |
N2O5 |
+ |
O3 |
2 |
NO3 |
+ |
O2 |
NH3 |
+ |
2 |
O2 |
HNO3 |
+ |
H2O |
4 |
NH3 |
+ |
5 |
O2 |
+ |
Pt |
+ |
4 |
NO |
+ |
6 |
H2O |
P |
+ |
3 |
O2 |
2 |
P2O3 |
P2O3 |
+ |
O2 |
P2O5 |
4 |
As |
+ |
3 |
O2 |
2 |
As2O3 |
4 |
Sb |
+ |
3 |
O2 |
2 |
Sb2O3 |
4 |
Bi |
+ |
3 |
O2 |
2 |
Bi2O3 |
As2S3 |
+ |
3 |
O2 |
2 |
As |
+ |
3 |
SO2 |
HOOH |
+ |
NaOCl(aq) |
1ΔO2 |
+ |
NaCl |
+ |
H2O |
+ |
1ΔO2 |
2 |
KI |
+ |
O3 |
+ |
H2O |
I2 |
+ |
2 |
KOH |
+ |
O2 |
2 |
NO2 |
+ |
O3 |
N2O5 |
+ |
O2 |
2 |
KOH |
+ |
5 |
O3 |
2 |
KO3 |
+ |
5 |
O2 |
+ |
H2O |
Se |
+ |
O2 |
SeO2 |
Te |
+ |
4 |
HNO3 |
TeO2 |
+ |
2 |
H2O |
+ |
4 |
NO2 |
2 |
Na2S2O3 |
+ |
I2 |
+ |
Starch |
Na2S4O6 |
+ |
2 |
NaI |
2 |
+ |
I2 |
+ |
Starch |
+ |
2 |
I– |
Na2SO3 |
+ |
S |
+ |
H2O |
Na2S2O3 |
[SO3]2– |
+ |
HOOH |
[SO4]2– |
+ |
H2O |
[SO3]2– |
+ |
S |
[HSO3]– |
+ |
I2 |
+ |
H2O |
[SO4]2– |
+ |
2 |
I– |
HSO3Cl |
+ |
HOOH |
+ |
HCl |
H2SeO3 |
+ |
HOOH |
+ |
H2O |
SO2 |
+ |
Cl2 |
SO2Cl2 |
Ag |
+ |
F2 |
AgF2 |
SO2 |
+ |
2 |
AgF2 |
SF2O2 |
+ |
2 |
AgF |
2 |
AgCl |
+ |
2 |
F2 |
2 |
AgF2 |
+ |
Cl2 |
5 |
HOOH |
+ |
2 |
[MnO4]– |
+ |
6 |
H+ |
5 |
O2 |
+ |
2 |
Mg2+ |
+ |
8 |
H2O |
HOOH |
+ |
Cl2 |
2 |
HCl |
+ |
O2 |
S |
+ |
3 |
F2 |
SF6 |
Se |
+ |
3 |
F2 |
SeF6 |
Te |
+ |
3 |
F2 |
TeF6 |
S |
+ |
4 |
CoF3 |
SF4 |
+ |
4 |
CoF2 |
2 |
Se |
+ |
Cl2 |
Se2Cl2 |
2 |
Se |
+ |
Br2 |
S2Br2 |
Se |
+ |
2 |
Br2 |
SeBr4 |
Se |
+ |
2 |
Cl2 |
SeCl4 |
Te |
+ |
2 |
Cl2 |
TeCl4 |
Te |
+ |
2 |
Br2 |
TeBr4 |
2 |
S |
+ |
5 |
F2 |
+ |
N2 |
SF4 |
+ |
SF6 |
Cl–S–S–Cl |
+ |
Cl2 |
2 |
SCl2 |
Cl2 |
+ |
3 |
F2 |
2 |
ClF3 |
UF4 |
+ |
F2 |
UF6 |
UF4 |
+ |
ClF3 |
UF6 |
+ |
ClF |
2 |
I– |
+ |
F2 |
I2 |
+ |
2 |
F– |
2 |
Br– |
+ |
F2 |
Br2 |
+ |
2 |
F– |
2 |
Cl– |
+ |
F2 |
Cl2 |
+ |
2 |
F– |
2 |
I– |
+ |
Cl2 |
I2 |
+ |
2 |
Cl– |
2 |
Br– |
+ |
Cl2 |
Br2 |
+ |
2 |
Cl– |
2 |
I– |
+ |
Br2 |
I2 |
+ |
2 |
Br– |
2 |
H2O |
+ |
2 |
F2 |
O2 |
+ |
4 |
HF |
4 |
I– |
+ |
O2 |
+ |
4 |
H+ |
2 |
I2 |
+ |
2 |
H2O |
8 |
NH3 |
+ |
3 |
F2 |
N2 |
+ |
6 |
[NH4]+ |
+ |
6 |
F– |
8 |
NH3 |
+ |
3 |
Cl2 |
N2 |
+ |
6 |
NH4Cl |
8 |
NH3 |
+ |
3 |
Br2 |
N2 |
+ |
6 |
[NH4]+ |
+ |
6 |
Br– |
2 |
I2O5 |
+ |
10 |
F2 |
4 |
IF5 |
+ |
5 |
O2 |
O2 |
+ |
F2 |
O2F2 |
Br2 |
+ |
2 |
O3 |
2 |
BrO2 |
+ |
O2 |
H2O |
+ |
F2 |
HOF |
+ |
HF |
2 |
ClO2 |
+ |
Na2O2 |
2 |
NaClO2 |
+ |
O2 |
10 |
Al |
+ |
6 |
NH4ClO4 |
+ |
Butyl Rubber |
5 |
Al2O3 |
+ |
6 |
HCl |
+ |
3 |
N2 |
+ |
9 |
H2O |
NaBrO3 |
+ |
F2 |
+ |
2 |
KOH |
NaBrO4 |
+ |
2 |
KF |
+ |
H2O |
[IO3]– |
+ |
Cl2 |
+ |
6 |
[OH]– |
+ |
5 |
H+ |
H5IO6 |
+ |
3 |
H2O |
+ |
2 |
Cl– |
3 |
I2 |
+ |
10 |
HNO3 |
6 |
HIO3 |
+ |
10 |
NO |
+ |
2 |
H2O |
I2 |
+ |
5 |
HOOH |
2 |
HIO3 |
+ |
4 |
H2O |
Cl2 |
+ |
F2 |
2 |
ClF |
Cl2 |
+ |
3 |
F2 |
2 |
ClF3 |
I2 |
+ |
Cl2 |
2 |
I–Cl |
I2 |
+ |
3 |
Cl2 |
I2Cl6 |
Br2 |
+ |
3 |
F2 |
+ |
N2 |
2 |
BrF3 |
Br2 |
+ |
5 |
F2 |
2 |
BrF5 |
I2 |
+ |
F2 |
2 |
I–F |
I2 |
+ |
5 |
F2 |
2 |
IF5 |
I2 |
+ |
7 |
F2 |
2 |
IF7 |
Br2 |
+ |
3 |
BrF5 |
5 |
BrF3 |
IF5 |
+ |
F2 |
IF7 |
I2 |
+ |
Br2 |
2 |
I–Br |
S |
+ |
6 |
ClF |
SF6 |
+ |
3 |
Cl2 |
2 |
AgCl |
+ |
2 |
ClF3 |
2 |
AgF2 |
+ |
2 |
ClF |
+ |
Cl2 |
SiO2 |
+ |
2 |
BrF5 |
SiF4 |
+ |
2 |
BrF3 |
+ |
O2 |
KI |
+ |
4 |
F2 |
IF7 |
+ |
KF |
O2 |
+ |
PtF6 |
[O–O]+ |
+ |
[PtF6]– |
Rn |
+ |
F2 |
RnF2 |
Kr |
+ |
F2 |
KrF2 |
Xe |
+ |
F2 |
XeF2 |
Xe |
+ |
2 |
F2 |
XeF4 |
Xe |
+ |
3 |
F2 |
XeF6 |
XeF2 |
+ |
2 |
HCl |
Cl2 |
+ |
2 |
HF |
+ |
Xe |
2 |
SF4 |
+ |
XeF4 |
2 |
SF6 |
+ |
Xe |
Pt |
+ |
3 |
XeF2 |
+ |
HF |
PtF6 |
+ |
3 |
Xe |
CH3I |
+ |
XeF2 |
CH3IF |
+ |
Xe |
Ar |
+ |
HF |
+ |
hν |
H–Ar–F |
CH4 |
+ |
2 |
Cl2 |
+ |
hν |
CH2Cl2 |
+ |
2 |
HCl |
CH4 |
+ |
3 |
Cl2 |
+ |
hν |
CHCl3 |
+ |
3 |
HCl |
CH4 |
+ |
4 |
Cl2 |
+ |
hν |
CCl4 |
+ |
4 |
HCl |
Xe |
+ |
2 |
PtF6 |
[XeF]+ [PtF6]– |
+ |
PtF5 |
2 |
H2C=CH2 |
+ |
O2 |
+ |
H2O |
+ |
PdCl2 |
+ |
CuCl2 |
2 |
CH3CHO |
H2C=CH2 |
+ |
PdCl2 |
+ |
H2O |
CH3CHO |
+ |
Pd |
+ |
2 |
HCl |
Pd |
+ |
2 |
CuCl2 |
PdCl2 |
+ |
2 |
CuCl |
4 |
CuCl |
+ |
O2 |
+ |
4 |
HCl |
4 |
CuCl2 |
+ |
2 |
H2O |
+ |
+ |
2 |
H2 |
+ |
O2 |
2 |
H2O |
+ |
Se |
+ |
+ |
+ |
RCOOH |
+ |
O3 |
+ |
Zn |
+ |
CH3COOH |
2 |
+ |
O3 |
+ |
Zn |
+ |
CH3COOH |
+ |
RCHO |
H2C=CH2 |
+ |
HClO |
2 |
H2C=CH2 |
+ |
O2 |
+ |
Ag |
2 |
4 |
H2C=CH2 |
+ |
2 |
O2 |
+ |
6 |
CH3COOH |
2 |
+ |
2 |
+ |
2 |
H2O |
2 |
H2C=CH2 |
+ |
O2 |
+ |
2 |
CH3COOH |
+ |
PdCl2 |
2 |
+ |
2 |
H2O |
H2C=CH2 |
+ |
Cl2 |
+ |
FeCl3 |
CH2ClCH2Cl |
2 |
H2C=CH2 |
+ |
4 |
HCl |
+ |
O2 |
+ |
CuCl2 |
2 |
CH2ClCH2Cl |
+ |
2 |
H2O |
2 |
+ |
O2 |
2 |
CH3CH2COOH |
4 |
CH4 |
+ |
2 |
Cl2 |
+ |
O2 |
4 |
CH3Cl |
+ |
2 |
H2O |
2 |
CH4 |
+ |
2 |
Cl2 |
+ |
O2 |
2 |
CH2Cl2 |
+ |
2 |
H2O |
4 |
CH4 |
+ |
6 |
Cl2 |
+ |
3 |
O2 |
4 |
CHCl3 |
+ |
6 |
H2O |
CH4 |
+ |
2 |
Cl2 |
+ |
O2 |
CCl4 |
+ |
2 |
H2O |
4 |
+ |
3 |
O2 |
2 |
+ |
2 |
(CH3)3COH |
CH3–CH=CH2 |
+ |
+ |
(CH3)3COH |
+ |
O2 |
2 |
+ |
O2 |
2 |
+ |
2 |
CO2 |
2 |
+ |
3 |
O2 |
2 |
+ |
2 |
H2O |
+ |
3 |
O2 |
+ |
V2O5 |
+ |
3 |
H2O |
2 |
+ |
9 |
O2 |
+ |
V2O5 |
2 |
+ |
4 |
CO2 |
+ |
4 |
H2O |
+ |
3 |
O2 |
+ |
CH3COOH |
+ |
2 |
H2O |
+ |
HNO3 |
+ |
4 |
H2 |
2 |
H2O |
+ |
2 |
Cl2 |
4 |
HCl |
+ |
O2 |
+ |
O2 |
HOOH |
+ |
+ |
O2 |
HOOH |
+ |
6 |
H2S |
+ |
3 |
O2 |
6 |
S |
+ |
6 |
H2O |
2 |
SO2 |
+ |
O2 |
+ |
V2O5 |
2 |
SO3 |
SO2 |
+ |
2 |
NOHSO4 |
+ |
2 |
H2O |
3 |
H2SO4(aq) |
+ |
2 |
NO |
2 |
S |
+ |
5 |
F2 |
S2F10 |
2 |
HI |
+ |
Cl2 |
+ |
H2SO4 |
2 |
HCl |
+ |
I2 |
H2N–NH2 |
+ |
2 |
I2 |
4 |
HI |
+ |
N2 |
4 |
MnO2 |
+ |
O2 |
+ |
12 |
KOH |
4 |
K3MnO4 |
+ |
6 |
H2O |
4 |
K3MnO4 |
+ |
O2 |
+ |
2 |
H2O |
4 |
K2MnO4 |
+ |
4 |
KOH |
2 |
CH3OH |
+ |
O2 |
+ |
Ag |
2 |
HCHO |
+ |
2 |
H2O |
+ |
HNO3 fuming |
HCN |
+ |
Cl2 |
+ |
H2O |
N≡C–Cl |
+ |
HCl |
2 |
+ |
O2 |
2 |
2 |
+ |
O2 |
2 |
+ |
O2 |
+ |
Ag |
+ |
2 |
H2O |
2 |
+ |
O2 |
2 |
2 |
CH3CH2OH |
+ |
O2 |
+ |
Ag |
2 |
CH3CHO |
+ |
2 |
H2O |
2 |
+ |
O2 |
2 |
2 |
CH3CHO |
+ |
O2 |
+ |
Cu2+ |
(CH3CO)2CO |
+ |
H2O |
2 |
CH3CHO |
+ |
O2 |
2 |
CH3COOH |
CH3CHO |
+ |
O2 |
+ |
CH3COOOH |
+ |
O2 |
+ |
Co2+ |
CH3COOH |
+ |
+ |
HCOOH |
+ |
CH3CH2COOH |
+ |
O2 |
+ |
Co2+ |
+ |
+ |
+ |
CH3CHO |
CH3COOH |
+ |
Cl2 |
CH2ClCOOH |
+ |
CHCl2COOH |
+ |
CCl3COOH |
+ |
HNO3 |
+ |
CO2 |
+ |
HNO3 |
+ |
NH3 |
+ |
O2 |
2 |
+ |
O2 |
+ |
+ |
+ |
3.5 |
O2 |
2 |
+ |
H2O |
+ |
2 |
O2 |
+ |
NOCl |
+ |
HCl |
+ |
hν |
+ |
HCl |
CH3–CH=CH2 |
+ |
CH3COOOH |
+ |
CH3COOH |
+ |
CH3COOH |
2 |
CH3–CH=CH2 |
+ |
O2 |
+ |
PdCl2 |
2 |
CH3–CH=CH2 |
+ |
O2 |
+ |
Cu2O |
+ |
H2O |
2 |
CH3–CH=CH2 |
+ |
2.5 |
O2 |
+ |
+ |
2 |
H2O |
2 |
+ |
2 |
HClO |
+ |
Ca(OH)2 |
2 |
+ |
CaCl2 |
+ |
2 |
H2O |
+ |
CH3COOOH |
+ |
CH3COOH |
2 |
+ |
2 |
Cl2 |
+ |
Ca(OH)2 |
2 |
+ |
CaCl2 |
+ |
2 |
H2O |
+ |
HOOH |
4 |
CH3–CH=CH2 |
+ |
6 |
NO |
+ |
Ag2O |
4 |
+ |
6 |
H2O |
+ |
N2 |
2 |
+ |
O2 |
2 |
(CH3)3COH |
+ |
O2 |
+ |
2 |
H2O |
2 |
+ |
3 |
O2 |
+ |
CrO3 |
2 |
+ |
2 |
H2O |
+ |
+ |
MnO2 |
+ |
H2SO4 |
2 |
+ |
9 |
O2 |
+ |
V2O5 |
2 |
+ |
4 |
CO2 |
+ |
4 |
H2O |
+ |
+ |
+ |
9 |
O2 |
+ |
V2O5 |
3 |
+ |
9 |
H2O |
2 |
+ |
7 |
O2 |
+ |
V2O5 |
2 |
+ |
8 |
H2O |
+ |
HOOH |
+ |
H2O |
+ |
H+ |
+ |
2.5 |
O2 |
+ |
V2O5 |
+ |
2 |
H2O |
CH3CN |
+ |
LiAlH4 |
+ |
4 |
[H] |
CH3CH2NH2 |
+ |
Fehling's Solution |
+ |
Cu2O |
+ |
Tollens' Reagent |
+ |
Ag |
Cu |
+ |
HOOH |
+ |
2 |
HCl(conc) |
Cu2+ |
+ |
2 |
Cl– |
+ |
2 |
H2O |
5 |
Fe2+ |
+ |
[MnO4]– |
+ |
8 |
H+ |
5 |
Fe3+ |
+ |
Mg2+ |
+ |
4 |
H2O |
5 |
[Fe(H2O)6]2+ |
+ |
[MnO4]– |
+ |
4 |
H2SO4(aq) |
+ |
2 |
H2O |
5 |
[Fe(H2O)6]3+ |
+ |
[Mn(H2O)6]2+ |
+ |
4 |
[SO4]2– |
6 |
Fe2+ |
+ |
[Cr2O7]2– |
+ |
14 |
H+ |
6 |
Fe3+ |
+ |
2 |
Cr3+ |
+ |
7 |
H2O |
6 |
[Fe(H2O)6]2+ |
+ |
[Cr2O7]2– |
+ |
7 |
H2SO4(aq) |
6 |
[Fe(H2O)6]3+ |
+ |
2 |
Cr3+ |
+ |
7 |
H2O |
+ |
7 |
[SO4]2– |
2 |
[Cr(OH)6]3– |
+ |
3 |
HOOH |
2 |
[CrO4]2– |
+ |
2 |
[OH]– |
+ |
8 |
H2O |
SO2 |
+ |
V2O5 |
SO3 |
+ |
VO2 |
2 |
VO2 |
+ |
O2 |
2 |
V2O5 |
5 |
[C2O4]2– |
+ |
2 |
[MnO4]– |
+ |
16 |
H+ |
+ |
Mg2+ |
10 |
CO2 |
+ |
2 |
Mg2+ |
+ |
8 |
H2O |
4 |
Mg2+ |
+ |
[MnO4]– |
+ |
8 |
H+ |
5 |
Mn3+ |
+ |
4 |
H2O |
[C2O4]2– |
+ |
2 |
Mn3+ |
2 |
CO2 |
+ |
2 |
Mg2+ |
+ |
2 |
[Ag(NH3)2]+ |
+ |
3 |
NaOH(aq) |
+ |
2 |
Ag |
+ |
4 |
NH3 |
+ |
2 |
H2O |
+ |
3 |
Na+ |
CH3CHO |
+ |
2 |
[Ag(NH3)2]+ |
+ |
3 |
NaOH(aq) |
[CH3COO]– |
+ |
2 |
Ag |
+ |
4 |
NH3 |
+ |
2 |
H2O |
+ |
3 |
Na+ |
2 |
[Cr(H2O)6]2+ |
+ |
3 |
HOOH |
+ |
10 |
NaOH(aq) |
2 |
[CrO4]2– |
+ |
10 |
Na+ |
+ |
20 |
H2O |
3 |
+ |
4 |
K2Cr2O7 |
+ |
32 |
H+ |
3 |
+ |
8 |
Cr3+ |
+ |
8 |
K+ |
+ |
22 |
H2O |
3 |
CH3CH2OH |
+ |
2 |
[Cr2O7]2– |
+ |
16 |
H+ |
3 |
CH3COOH |
+ |
4 |
Cr3+ |
+ |
11 |
H2O |
Cu |
+ |
2 |
[NO3]– |
+ |
4 |
H+ |
Cu2+ |
+ |
2 |
NO2 |
+ |
2 |
H2O |
4 |
Ag |
+ |
O2 |
+ |
8 |
[CN]– |
+ |
2 |
H2O |
4 |
[Ag(CN)2]– |
+ |
4 |
[OH]– |
2 |
KI |
+ |
Br2 |
2 |
KBr |
+ |
I2 |
2 |
HBr |
+ |
H2SO4 |
Br2 |
+ |
SO2 |
+ |
2 |
H2O |
4 |
NaBr |
+ |
3 |
H2SO4 |
2 |
HBr |
+ |
2 |
Na2SO4 |
+ |
Br2 |
+ |
SO2 |
+ |
2 |
H2O |
2 |
NaI |
+ |
H2SO4 |
2 |
HI |
+ |
Na2SO4 |
2 |
HI |
+ |
H2SO4 |
I2 |
+ |
SO2 |
+ |
2 |
H2O |
4 |
HI |
+ |
SO2 |
2 |
I2 |
+ |
S |
+ |
2 |
H2O |
2 |
HI |
+ |
S |
I2 |
+ |
H2S |
NaI |
+ |
H2SO4 |
HI |
+ |
I2 |
+ |
SO2 |
+ |
S |
+ |
H2S |
2 |
CH3OH |
+ |
3 |
O2 |
2 |
CO2 |
+ |
4 |
H2O |
Ba |
+ |
2 |
H2O |
Ba2+ |
+ |
2 |
[OH]– |
+ |
H2 |
CS2 |
+ |
2 |
H2O |
CO2 |
+ |
2 |
H2S |
SO2 |
+ |
Cl2 |
+ |
2 |
H2O |
H2SO4 |
+ |
2 |
HCl |
Mg |
+ |
2 |
HCl |
MgCl2 |
+ |
H2 |
2 |
H2O |
+ |
2 |
Cl2 |
+ |
hν |
O2 |
+ |
4 |
HCl |
2 |
+ |
9 |
O2 |
6 |
CO2 |
+ |
8 |
H2O |
2 |
+ |
9 |
O2 |
6 |
CO2 |
+ |
8 |
H2O |
3 |
Cu |
+ |
2 |
[NO3]– |
+ |
8 |
H+ |
+ |
H2O |
3 |
Cu2+ |
+ |
2 |
NO |
+ |
4 |
H2O |
2 |
NaBr |
+ |
Cl2 |
Br2 |
+ |
2 |
NaCl |
Ti |
+ |
O2 |
TiO2 |
Ti |
+ |
C |
TiC |
2 |
Ti |
+ |
N2 |
2 |
TiN |
+ |
4 |
O2 |
3 |
CO2 |
+ |
3 |
H2O |
2 |
CH3CHO |
+ |
5 |
O2 |
4 |
CO2 |
+ |
4 |
H2O |
2 |
NO |
+ |
10 |
I– |
+ |
12 |
H+ |
2 |
[NH4]+ |
+ |
5 |
I2 |
+ |
2 |
H2O |
SO2 |
+ |
Cl2 |
+ |
2 |
H2O |
[SO4]2– |
+ |
2 |
Cl– |
+ |
4 |
H+ |
SO2 |
+ |
Cl2 |
+ |
2 |
H2O |
H2SO4 |
+ |
2 |
HCl |
2 |
KI |
+ |
Cl2 |
+ |
H2O |
I2 |
+ |
2 |
KCl |
+ |
3 |
O2 |
4 |
CO2 |
+ |
2 |
H2O |
4 |
Fe |
+ |
3 |
O2 |
+ |
S |
2 |
Fe2O3 |
+ |
S |
2 |
NaI |
+ |
Br2 |
I2 |
+ |
2 |
NaBr |
6 |
Fe2+ |
+ |
[ClO3]– |
+ |
6 |
H+ |
6 |
Fe3+ |
+ |
Cl– |
+ |
3 |
H2O |
+ |
[O] |
+ |
H2O |
+ |
[O] |
+ |
H2O |
+ |
[O] |
+ |
H2O |
+ |
Fehling's Solution |
+ |
Cu2O |
+ |
Tollens' Reagent |
+ |
Ag |
3 |
+ |
[Cr2O7]2– |
+ |
8 |
H+ |
3 |
+ |
2 |
Cr3+ |
+ |
4 |
H2O |
+ |
[O] |
2 |
H2S |
+ |
3 |
O2 |
2 |
SO2 |
+ |
2 |
H2O |
N2 |
+ |
O2 |
2 |
NO |
4 |
NO2 |
+ |
O2 |
+ |
2 |
H2O |
4 |
HNO3(g) |
CH3CH2OH |
+ |
3 |
O2 |
2 |
CO2 |
+ |
3 |
H2O |
+ |
23 |
O2 |
15 |
CO2 |
+ |
16 |
H2O |
2 |
CH4 |
+ |
3 |
O2 |
2 |
CO |
+ |
4 |
H2O |
+ |
[O] |
+ |
H2O |
+ |
Fehling's Solution |
+ |
Cu2O |
CH3CH2OH |
+ |
[O] |
CH3CHO |
+ |
H2O |
CH3CH2OH |
+ |
2 |
[O] |
CH3COOH |
+ |
H2O |
+ |
[O] |
+ |
H2O |
CH4 |
+ |
O2 |
C |
+ |
2 |
H2O |
CH3Cl |
+ |
Cl2 |
+ |
hν |
CH2Cl2 |
+ |
HCl |
2 |
+ |
9 |
O2 |
8 |
CO |
+ |
10 |
H2O |
CHCl3 |
+ |
Cl2 |
+ |
hν |
CCl4 |
+ |
HCl |
+ |
[O] |
+ |
H2O |
+ |
2 |
[O] |
+ |
H2O |
+ |
12.5 |
O2 |
8 |
CO2 |
+ |
9 |
H2O |
+ |
8.5 |
O2 |
8 |
CO |
+ |
9 |
H2O |
+ |
3 |
+ |
+ |
3 |
+ |
3 |
+ |
+ |
3 |
+ |
4 |
+ |
+ |
4 |
+ |
[O] |
+ |
[O] |
2 |
[Co(NH3)6]2+ |
+ |
HOOH |
2 |
[Co(NH3)6]3+ |
+ |
2 |
[OH]– |
3 |
Zn |
+ |
K2Cr2O7 |
+ |
14 |
H+ |
3 |
Zn2+ |
+ |
2 |
Cr3+ |
+ |
2 |
K+ |
+ |
7 |
H2O |
3 |
CH3CHO |
+ |
K2Cr2O7 |
+ |
8 |
H+ |
3 |
CH3COOH |
+ |
2 |
Cr3+ |
+ |
2 |
K+ |
+ |
4 |
H2O |
6 |
Fe2+ |
+ |
K2Cr2O7 |
+ |
14 |
H+ |
6 |
Fe3+ |
+ |
2 |
Cr3+ |
+ |
2 |
K+ |
+ |
7 |
H2O |
[VO]2+ |
+ |
e– |
+ |
2 |
H+ |
V3+ |
+ |
H2O |
5 |
+ |
2 |
KMnO4 |
+ |
6 |
H+ |
10 |
CO2 |
+ |
2 |
Mg2+ |
+ |
8 |
H2O |
+ |
2 |
K+ |
3 |
HCl(conc) |
+ |
HNO3 |
Cl2 |
+ |
NOCl |
+ |
2 |
H2O |
Aqua Regia |
Cl2 |
+ |
NOCl |
+ |
2 |
H2O |
Au |
+ |
Aqua Regia |
+ |
Cl– |
[AuCl4]– |
+ |
NO |
+ |
2 |
H2O |
3 |
+ |
3 |
HOOH |
+ |
H2SO4 |
+ |
3 |
H2O |
CH4 |
+ |
2 |
Cl2 |
C |
+ |
4 |
HCl |
© Mark R. Leach 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com