
Interactions | Reactions | Processes
Classified as: Functional Group Interconvertion
Functional Group Interconvertion (FGI) is a concept associated with "RetroSynthetic Analysis", RSA, a rational approach to the designing synthetic routes to large organic structures, usually of pharmaceutical interest. The oxidation of a secondary alcohol to a ketone is an FGI.
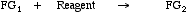
+ |
H2O |
+ |
H+(aq) |
RCHO |
+ |
2 |
R–OH |
(CH3CO)2CO |
+ |
2 |
[OH]– |
2 |
[CH3COO]– |
+ |
H2O |
(CH3CO)2CO |
+ |
2 |
R–OH |
+ |
CH3COOR |
+ |
CH3COOH |
(CH3CO)2CO |
+ |
NH3 |
CH3CONH2 |
+ |
CH3COOH |
(CH3CO)2CO |
+ |
LiAlH4 |
+ |
8 |
[H] |
+ |
2 |
CH3CH2OH |
+ |
H2O |
RCOCl |
+ |
H2O |
RCOOH |
+ |
HCl |
RCOCl |
+ |
R–OH |
+ |
+ |
RCOCl |
+ |
2 |
NH3 |
+ |
NH4Cl |
RCOCl |
+ |
LiAlH4 |
+ |
RCH2OH |
RCOCl |
+ |
Li[Al(OtBu)3H] 1.0M in THF |
+ |
RCHO |
RCOCl |
+ |
H2 |
+ |
Pd on BaSO4 |
RCHO |
+ |
HCl |
RCH2OH |
+ |
RCHO |
RCH2OH |
+ |
CrO3 |
RCOOH |
+ |
RCOOH |
+ |
R–OH |
+ |
H2O |
+ |
H2SO4 |
CH3–CH=CH2 |
+ |
H2O |
+ |
H2SO4 |
R–I |
+ |
RO– |
+ |
NaI |
(CH3)3COH |
+ |
HCl(conc) |
+ |
H2O |
(CH3)3COH |
+ |
HBr |
(CH3)3CBr |
+ |
H2O |
(CH3)3COH |
+ |
HI |
(CH3)3CI |
+ |
H2O |
R–OH |
+ |
SOCl2 |
RCl |
R–OH |
+ |
PBr3 |
RBr |
RCHO |
+ |
CrO3 |
RCOOH |
RCHO |
+ |
NaBH4 |
RCH2OH |
RCHO |
+ |
HCN |
RCHO |
+ |
H2N–NH2 |
+ |
OH–(aq) |
R–CH3 |
+ |
N2 |
RCHO |
+ |
Zn(Hg) |
+ |
H+(aq) |
R–CH3 |
+ |
Zn2+ |
RCHO |
+ |
2 |
R–OH |
+ |
+ |
2 |
H2O |
RCHO |
+ |
+ |
+ |
2 |
H2O |
RCHO |
+ |
+ |
+ |
2 |
H2O |
+ |
H2 |
+ |
Ni/H2 Raney Nickel |
R–CH3 |
RCHO |
+ |
RCHO |
+ |
R–NH2 |
+ |
H2O |
RCH2CHO |
+ |
Br2 |
+ |
CH3COOH |
+ |
HBr |
+ |
[OH]– |
RCOO– |
+ |
NH3 |
+ |
LiAlH4 |
+ |
RCH2NH2 |
+ |
SOCl2 |
R–CN |
+ |
HCl |
+ |
SO2 |
CH4 |
+ |
Cl2 |
+ |
hν |
CH3Cl |
+ |
HCl |
+ |
HCl |
+ |
HBr |
+ |
HI |
+ |
Cl2 |
+ |
Br2 |
+ |
H2 |
+ |
Pd on C |
R–CH3 |
+ |
+ |
R–C≡C–R |
+ |
HCl |
+ |
HCl |
R–C≡C–R |
+ |
Cl2 |
+ |
Cl2 |
R–C≡C–H |
+ |
HgSO4 |
+ |
H+(aq) |
R–C≡C–H |
+ |
+ |
HOOH |
+ |
NaOH |
RCH2CHO |
2 |
+ |
BF3←THF in THF |
+ |
R–C≡C–R |
+ |
H2 |
+ |
Pd + Pb on CaCO3 Lindlar Cat. |
R–C≡C–R |
+ |
Li |
+ |
NH3 |
+ |
R–NH2 |
+ |
R–OH |
R–NH2 |
+ |
10 |
CH3I |
+ |
Ag2O |
+ |
(CH3)3N |
+ |
HNO2 |
+ |
H+ |
+ |
H2O |
+ |
2 |
H2O |
+ |
KMnO4 |
+ |
2 |
H2 |
+ |
Pd on C |
+ |
H2O |
+ |
Cl+ |
+ |
H+ |
+ |
Cl+ / [AlCl4]– |
+ |
H+ |
+ |
[AlCl4]– |
+ |
Br+ |
+ |
H+ |
+ |
Br+ [FeBr4]– |
+ |
H+ |
+ |
[FeBr4]– |
+ |
I2 |
+ |
CuCl2 |
+ |
[NO2]+ |
+ |
H+ |
+ |
HNO3 |
+ |
H2SO4 |
+ |
H2O |
+ |
H2S2O7 |
+ |
H2SO4 |
+ |
H2S2O7 |
+ |
H2O |
+ |
H2SO4 |
+ |
HSO3Cl |
+ |
H2O |
+ |
KMnO4 |
+ |
OH–(aq) |
+ |
RCOOH |
RCl |
+ |
AlCl3 |
R+ [AlCl4]– |
RCOCl |
+ |
AlCl3 |
+ |
CuCl |
+ |
N2 |
+ |
Cu+ |
+ |
CuBr |
+ |
N2 |
+ |
Cu+ |
+ |
NaI |
+ |
N2 |
+ |
Na+ |
+ |
H2O |
+ |
N2 |
+ |
H+ |
+ |
H3PO2 |
+ |
[H] |
+ |
N2 |
+ |
NaOH |
+ |
Na+ |
+ |
[HSO3]– |
RCOOH |
+ |
Base– Proton abstractor |
RCOO– |
+ |
Base-H Protonated base |
RCOOH |
+ |
LiAlH4 |
RCH2OH |
2 |
RCOOH |
+ |
B2H6 |
2 |
RCH2OH |
RCOOH |
+ |
SOCl2 |
RCOCl |
+ |
SO2 |
+ |
HCl |
RCOCl |
+ |
RCOO– |
(RCOO)2O |
+ |
Cl– |
RCOO– |
+ |
RCl |
+ |
Cl– |
RCOOH |
+ |
CH2N2 |
+ |
N2 |
+ |
HCl |
+ |
H+(aq) |
+ |
[OH]– |
RCOO– |
+ |
R–OH |
+ |
NH3 |
+ |
R–OH |
+ |
LiAlH4 |
RCH2OH |
+ |
R–OH |
+ |
RCHO |
+ |
R–OH |
RCl |
+ |
Mg |
RMgCl |
RBr |
+ |
Mg |
RMgBr |
CH3I |
+ |
Mg |
CH3MgI |
RMgCl |
+ |
H2O |
R–H |
+ |
Mg2+ |
+ |
[OH]– |
CH3MgI |
+ |
H2O |
CH4 |
+ |
Mg2+ |
+ |
[OH]– |
+ |
I– |
+ |
Base– Proton abstractor |
+ |
Cl– |
+ |
Base– Proton abstractor |
+ |
Br– |
+ |
Base– Proton abstractor |
+ |
HI |
+ |
NaOH |
+ |
NaBH4 |
R2CHOH |
+ |
H2N–NH2 |
+ |
OH–(aq) |
+ |
N2 |
+ |
HCN |
+ |
+ |
+ |
2 |
H2O |
+ |
+ |
H2O |
+ |
H2 |
+ |
Ni/H2 Raney Nickel |
+ |
+ |
(C6H5)3P=O |
+ |
R–NH2 |
+ |
H2O |
+ |
NH3 |
+ |
H2O |
+ |
Br2 |
+ |
CH3COOH |
+ |
HBr |
+ |
Zn(Hg) |
+ |
H+(aq) |
R–CN |
+ |
H+(aq) |
RCOOH |
+ |
[NH4]+ |
R–CN |
+ |
LiAlH4 |
+ |
4 |
[H] |
RCH2NH2 |
R–CN |
+ |
RCHO |
+ |
2 |
Fe |
+ |
6 |
H+ |
+ |
2 |
H2O |
+ |
2 |
Fe3+ |
+ |
3 |
Sn |
+ |
6 |
H+ |
+ |
3 |
Sn2+ |
+ |
2 |
H2O |
RBr |
+ |
Li |
RLi |
+ |
LiBr |
RLi |
+ |
CuBr |
R2CuLi |
+ |
LiBr |
+ |
[OH]– |
+ |
H2O |
RCOCl |
+ |
+ |
Cl– |
+ |
RBr |
+ |
Br– |
+ |
(KSO3)2NO |
+ |
NaBH4 |
+ |
2 |
[H] |
R2S |
+ |
R–I |
[R3S]+ |
+ |
I– |
+ |
CH3I |
R2S |
+ |
+ |
[R–S]– |
+ |
R–I |
R2S |
+ |
I– |
2 |
[R–S]– |
+ |
Br2 |
R–S–S–R |
+ |
2 |
Br– |
[R2OH]+ |
+ |
Br– |
RBr |
+ |
R–OH |
+ |
NaH |
+ |
+ |
H2 |
+ |
Na+ |
R–CH2–Br |
+ |
[(C6H5)3P–CH2R]+ |
+ |
Br– |
[(C6H5)3P–CH2R]+ |
+ |
NaH |
+ |
H2 |
RBr |
+ |
(CH3CH2O)3P |
+ |
CH3CH2Br |
R–CH2–Br |
+ |
(CH3CH2O)3P |
+ |
CH3CH2Br |
+ |
(CH3CH2O)3P |
+ |
CH3CH2Br |
R–NO2 |
+ |
2 |
(CH3CH2O)3P |
R–N |
+ |
2 |
(CH3CH2O)3P=O |
+ |
[OH]– |
+ |
H2S |
+ |
[OH]– |
+ |
NH3 |
+ |
LiAlH4 |
+ |
2 |
[H] |
CH3CH2OH |
+ |
[OH]– |
+ |
H2O |
+ |
SO2 |
+ |
Br2 |
(CH3)3CBr |
+ |
HBr |
+ |
Cl2 |
+ |
+ |
HCl |
+ |
Cl2 |
+ |
HCl |
+ |
3 |
H2 |
+ |
Rh on Carbon |
RCHO |
+ |
R–OH |
+ |
+ |
H2O |
+ |
R–OH |
+ |
+ |
H2O |
+ |
2 |
R–OH |
+ |
+ |
2 |
H2O |
+ |
H2O |
+ |
H2O |
+ |
H+(aq) |
+ |
CH3COOH |
RCOOH |
+ |
SF4 |
R–CF3 |
+ |
SO2 |
R–OH |
+ |
SF4 |
R–F |
+ |
SO2 |
CH4 |
+ |
2 |
Cl2 |
+ |
hν |
CH2Cl2 |
+ |
2 |
HCl |
CH4 |
+ |
3 |
Cl2 |
+ |
hν |
CHCl3 |
+ |
3 |
HCl |
CH4 |
+ |
4 |
Cl2 |
+ |
hν |
CCl4 |
+ |
4 |
HCl |
+ |
2 |
[Ag(NH3)2]+ |
+ |
3 |
NaOH(aq) |
+ |
2 |
Ag |
+ |
4 |
NH3 |
+ |
2 |
H2O |
+ |
3 |
Na+ |
CH3CHO |
+ |
2 |
[Ag(NH3)2]+ |
+ |
3 |
NaOH(aq) |
[CH3COO]– |
+ |
2 |
Ag |
+ |
4 |
NH3 |
+ |
2 |
H2O |
+ |
3 |
Na+ |
+ |
[OH]– |
+ |
HBr |
RCOOH |
+ |
2 |
LiCH3 |
© Mark R. Leach 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com