
Interactions | Reactions | Processes
Classified as: Proton transfer: Brønsted reaction
The Brønsted acid/base protonation reaction involves the transfer of a proton from a Lewis base complex to a Lewis base with a higher proton affinity. The process is a special case nucleophilic substitution (at an H+ centre).
This analysis is explained in detail in the Chemogenesis webbook.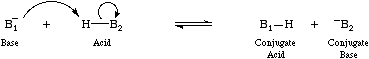
NaNO2 |
+ |
H+ |
HNO2 |
+ |
Na+ |
RCOOH |
+ |
Base– Proton abstractor |
RCOO– |
+ |
Base-H Protonated base |
+ |
HBr |
R–OH |
+ |
RBr |
RMgCl |
+ |
H2O |
R–H |
+ |
Mg2+ |
+ |
[OH]– |
CH3MgI |
+ |
H2O |
CH4 |
+ |
Mg2+ |
+ |
[OH]– |
+ |
I– |
+ |
Base– Proton abstractor |
+ |
Cl– |
+ |
Base– Proton abstractor |
+ |
Br– |
+ |
Base– Proton abstractor |
+ |
HI |
+ |
NaOH |
+ |
[OH]– |
+ |
H2O |
+ |
NaH |
+ |
+ |
H2 |
+ |
Na+ |
[(C6H5)3P–CH2R]+ |
+ |
NaH |
+ |
H2 |
+ |
[OH]– |
+ |
H2O |
+ |
HBF4(aq) |
+ |
H2O |
+ |
+ |
[BF4]– |
H+ |
+ |
H+ |
+ |
H2C=CH2 |
[CH3CH2]+ |
H+ |
+ |
CH3CH2OH |
HNO3 |
+ |
2 |
H2SO4 |
[NO2]+ |
+ |
2 |
[HSO4]– |
+ |
[H3O]+ |
NH3 |
+ |
H2O |
[NH4]+ |
+ |
[OH]– |
NH3(aq) |
+ |
H2O |
[NH4]+ |
+ |
[OH]– |
.880 NH3 |
+ |
H2O |
[NH4]+ |
+ |
[OH]– |
+ |
H2O |
+ |
CH3OH |
CuO |
+ |
H2SO4(aq) |
CuSO4 |
+ |
H2O |
CaC2 |
+ |
2 |
H2O |
HC≡CH |
+ |
Ca(OH)2 |
+ |
2 |
Base– Proton abstractor |
R–C≡C–R |
+ |
2 |
Base-H Protonated base |
+ |
2 |
Cl– |
+ |
NaNH2 |
+ |
NH3 |
R–C≡C–R |
+ |
NH3 |
+ |
NaCl |
R–C≡C–H |
+ |
RLi |
R–C≡C– |
+ |
Li+ |
+ |
R–H |
RLi |
+ |
H2O |
R–H |
+ |
Li+ |
+ |
[OH]– |
H+ |
+ |
[R–S–CH2]– |
R–S–CH3 |
R–S–CH3 |
+ |
Base– Proton abstractor |
[R–S–CH2]– |
+ |
Base-H Protonated base |
+ |
+ |
Li+ |
+ |
+ |
Base– Proton abstractor |
+ |
Base-H Protonated base |
+ |
Base– Proton abstractor |
+ |
Base-H Protonated base |
R–H |
+ |
Base– Proton abstractor |
R– |
+ |
Base-H Protonated base |
H+ |
+ |
H+ |
+ |
H+ |
+ |
+ |
H2O |
+ |
Li+ |
+ |
[OH]– |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
3 |
H+ |
+ |
3 |
3 |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
H+ |
+ |
Li3N |
+ |
3 |
H2O |
3 |
LiOH |
+ |
NH3 |
Na2O2 |
+ |
2 |
H2O |
HOOH |
+ |
2 |
NaOH |
Na2O2 |
+ |
2 |
HCl |
HOOH |
+ |
2 |
NaCl |
2 |
K2O |
+ |
2 |
H2O |
HOOH |
+ |
2 |
KOH |
+ |
O2 |
CO2 |
+ |
2 |
[OH]– |
[CO3]2– |
+ |
H2O |
LiH |
+ |
H2O |
LiOH |
+ |
H2 |
NaH |
+ |
H2O |
NaOH |
+ |
H2 |
KH |
+ |
H2O |
KOH |
+ |
H2 |
RbH |
+ |
H2O |
RbOH |
+ |
H2 |
CsH |
+ |
H2O |
CsOH |
+ |
H2 |
CH3CH2OH |
+ |
KOH |
CH3CH2O– |
+ |
K+ |
+ |
H2O |
2 |
NaOH |
+ |
H2S |
Na2S |
+ |
2 |
H2O |
2 |
NaCl |
+ |
H2SO4 |
2 |
HCl |
+ |
Na2SO4 |
[CO3]2– |
+ |
CO2 |
+ |
H2O |
+ |
H2O |
2 |
[HCO3]– |
LiOH |
+ |
H2S |
LiSH |
+ |
H2O |
NaOH |
+ |
H2S |
NaSH |
+ |
H2O |
KOH |
+ |
H2S |
KSH |
+ |
H2O |
RbOH |
+ |
H2S |
RbSH |
+ |
H2O |
CsOH |
+ |
H2S |
CsSH |
+ |
H2O |
2 |
LiOH |
+ |
H2S |
Li2S |
+ |
2 |
H2O |
2 |
KOH |
+ |
H2S |
K2S |
+ |
2 |
H2O |
2 |
RbOH |
+ |
H2S |
Rb2S |
+ |
2 |
H2O |
2 |
CsOH |
+ |
H2S |
Cs2S |
+ |
2 |
H2O |
Li2S |
+ |
H2O |
LiSH |
+ |
LiOH |
Na2S |
+ |
H2O |
NaSH |
+ |
NaOH |
K2S |
+ |
H2O |
KSH |
+ |
KOH |
Rb2S |
+ |
H2O |
RbSH |
+ |
RbOH |
Cs2S |
+ |
H2O |
CsSH |
+ |
CsOH |
2 |
LiNH2 |
+ |
NH3 |
LiNH2 |
+ |
NH3 |
Be3N2 |
+ |
6 |
H2O |
3 |
Be(OH)2 |
+ |
2 |
NH3 |
Mg3N2 |
+ |
6 |
H2O |
3 |
Mg(OH)2 |
+ |
2 |
NH3 |
Ca3N2 |
+ |
6 |
H2O |
3 |
Ca(OH)2 |
+ |
2 |
NH3 |
Sr3N2 |
+ |
6 |
H2O |
3 |
Sr(OH)2 |
+ |
2 |
NH3 |
Ba3N2 |
+ |
6 |
H2O |
3 |
Ba(OH)2 |
+ |
2 |
NH3 |
Ca2+ |
+ |
2 |
[HCO3]– |
+ |
Ca(OH)2 |
2 |
CaCO3 |
+ |
2 |
H2O |
BeH2 |
+ |
2 |
H2O |
Be(OH)2 |
+ |
2 |
H2 |
MgH2 |
+ |
2 |
H2O |
Mg(OH)2 |
+ |
2 |
H2 |
CaH2 |
+ |
2 |
H2O |
Ca(OH)2 |
+ |
2 |
H2 |
SrH2 |
+ |
2 |
H2O |
Sr(OH)2 |
+ |
2 |
H2 |
BaH2 |
+ |
2 |
H2O |
Ba(OH)2 |
+ |
2 |
H2 |
BeH2 |
+ |
2 |
H+ |
Be2+ |
+ |
2 |
H2 |
MgH2 |
+ |
2 |
H+ |
Mg2+ |
+ |
2 |
H2 |
CaH2 |
+ |
2 |
H+ |
Ca2+ |
+ |
2 |
H2 |
SrH2 |
+ |
2 |
H+ |
Sr2+ |
+ |
2 |
H2 |
BaH2 |
+ |
2 |
H+ |
Ba2+ |
+ |
2 |
H2 |
CaF2 |
+ |
H2SO4 |
2 |
HF |
+ |
CaSO4 |
Be2C |
+ |
4 |
H2O |
2 |
Be(OH)2 |
+ |
CH4 |
MgC2 |
+ |
2 |
H2O |
HC≡CH |
+ |
Mg(OH)2 |
SrC2 |
+ |
2 |
H2O |
HC≡CH |
+ |
Sr(OH)2 |
BaC2 |
+ |
2 |
H2O |
HC≡CH |
+ |
Ba(OH)2 |
4 |
H+ |
+ |
C4– |
CH4 |
D2 |
+ |
H2 |
+ |
[OH]– |
HD |
+ |
DOH |
Mg2Si |
+ |
2 |
H2SO4 |
SiH4 |
+ |
2 |
MgSO4 |
NH3 |
+ |
HCl |
NH4Cl |
NH4Cl |
NH3 |
+ |
HCl |
Ca3P2 |
+ |
6 |
H2O |
2 |
PH3 |
+ |
3 |
Ca(OH)2 |
Cs3As |
+ |
3 |
H2O |
H3As |
+ |
3 |
CsOH |
K3As |
+ |
3 |
H2O |
H3As |
+ |
3 |
KOH |
Li3As |
+ |
3 |
H2O |
H3As |
+ |
3 |
LiOH |
Na3As |
+ |
3 |
H2O |
H3As |
+ |
3 |
NaOH |
Rb3As |
+ |
3 |
H2O |
H3As |
+ |
3 |
RbOH |
Cs3Sb |
+ |
3 |
H2O |
H3Sb |
+ |
3 |
CsOH |
K3Sb |
+ |
3 |
H2O |
H3Sb |
+ |
3 |
KOH |
Li3Sb |
+ |
3 |
H2O |
H3Sb |
+ |
3 |
LiOH |
Na3Sb |
+ |
3 |
H2O |
H3Sb |
+ |
3 |
NaOH |
Rb3Sb |
+ |
3 |
H2O |
H3Sb |
+ |
3 |
RbOH |
H2N–OH |
+ |
HCl |
2 |
NH3 |
[NH4]+ |
+ |
[H2N]– |
3 |
HN3 |
+ |
2 |
Na |
2 |
NaN3 |
+ |
NH3 |
+ |
N2 |
N2O3 |
+ |
2 |
H2SO4 |
2 |
[NO]+ |
+ |
2 |
[HSO4]– |
+ |
H2O |
Na2CO3 |
+ |
SO2 |
+ |
H2O |
Na2SO3 |
+ |
CO2 |
Na2SO3 |
+ |
2 |
HCl |
+ |
H2O |
SO2 |
+ |
2 |
NaCl |
+ |
H2O |
+ |
H+ |
[HSO3]– |
+ |
SO2 |
+ |
2 |
Na+ |
Ag2CO3 |
+ |
2 |
HF |
2 |
AgF |
+ |
CO2 |
+ |
H2O |
K2CO3 |
+ |
4 |
HF |
2 |
K[HF2] |
+ |
CO2 |
+ |
H2O |
HCl |
+ |
H2O |
[H3O]+ |
+ |
Cl– |
2 |
HF |
H+ |
+ |
[FHF]– |
CH3CH2OH |
+ |
H2SO4 |
H2C=CH2 |
+ |
H2O |
+ |
H2SO4 |
CH3–CH=CH2 |
+ |
H2O |
(CH3)3COH |
+ |
H2SO4 |
+ |
H2O |
+ |
+ |
2 |
Na |
+ |
CH3CH2OH |
+ |
2 |
NaBr |
+ |
H2 |
+ |
2 |
Base– Proton abstractor |
[H3]+ |
+ |
Base: Proton abstractor |
+ |
H2 |
H+ |
+ |
H2 |
[H3]+ |
Na2[B4O5(OH)4] .8H2O |
+ |
2 |
HCl(aq) |
4 |
H3BO3 |
+ |
2 |
NaCl |
+ |
5 |
H2O |
2 |
H+ |
+ |
Ca3(PO4)2 |
+ |
3 |
H2SO4 |
2 |
H3PO4 |
+ |
3 |
CaSO4 |
NaOCl(aq) |
+ |
2 |
HCl |
Cl2 |
+ |
NaCl |
+ |
H2O |
NaOH |
+ |
HBr |
NaBr |
+ |
H2O |
NH3 |
+ |
HNO3 |
NH4NO3 |
HCOOH |
+ |
NaOH |
HCOONa |
+ |
H2O |
+ |
H2SO4 |
+ |
Na2SO4 |
+ |
H+ |
+ |
H2O |
CH3NH2 |
+ |
H2O |
[CH3NH3]+ |
+ |
[OH]– |
[Fe(H2O)6]2+ |
+ |
2 |
NaOH(aq) |
Fe(H2O)4(OH)2 |
+ |
2 |
Na+ |
+ |
2 |
H2O |
[Fe(H2O)6]2+ |
+ |
6 |
NaOH(aq) |
Fe(H2O)4(OH)2 |
+ |
2 |
Na+ |
+ |
2 |
H2O |
+ |
4 |
NaOH(aq) |
[Fe(H2O)6]2+ |
+ |
2 |
NH3(aq) |
Fe(H2O)4(OH)2 |
+ |
2 |
[NH4]+ |
[Fe(H2O)6]2+ |
+ |
2 |
NH3(aq) |
Fe(H2O)4(OH)2 |
+ |
2 |
[NH4]+ |
[Co(H2O)6]2+ |
+ |
2 |
NaOH(aq) |
Co(H2O)4(OH)2 |
+ |
2 |
Na+ |
+ |
2 |
H2O |
[Co(H2O)6]2+ |
+ |
6 |
NaOH(aq) |
Co(H2O)4(OH)2 |
+ |
2 |
Na+ |
+ |
2 |
H2O |
+ |
4 |
NaOH(aq) |
[Co(H2O)6]2+ |
+ |
2 |
NH3(aq) |
Co(H2O)4(OH)2 |
+ |
2 |
[NH4]+ |
[Cu(H2O)6]2+ |
+ |
2 |
NaOH(aq) |
Cu(H2O)4(OH)2 |
+ |
2 |
Na+ |
+ |
2 |
H2O |
[Cu(H2O)6]2+ |
+ |
6 |
NaOH(aq) |
Cu(H2O)4(OH)2 |
+ |
2 |
H2O |
+ |
2 |
Na+ |
+ |
4 |
NaOH(aq) |
[Cu(H2O)6]2+ |
+ |
2 |
NH3(aq) |
Cu(H2O)4(OH)2 |
+ |
2 |
[NH4]+ |
[Fe(H2O)6]3+ |
+ |
3 |
NaOH(aq) |
Fe(H2O)3(OH)3 |
+ |
3 |
H2O |
+ |
3 |
Na+ |
[Fe(H2O)6]3+ |
+ |
6 |
NaOH(aq) |
Fe(H2O)3(OH)3 |
+ |
3 |
H2O |
+ |
3 |
Na+ |
+ |
3 |
NaOH(aq) |
[Fe(H2O)6]3+ |
+ |
3 |
NH3(aq) |
Fe(H2O)3(OH)3 |
+ |
3 |
[NH4]+ |
[Fe(H2O)6]3+ |
+ |
6 |
NH3(aq) |
Fe(H2O)3(OH)3 |
+ |
3 |
[NH4]+ |
+ |
3 |
NH3 |
2 |
[Fe(H2O)6]3+ |
+ |
3 |
[CO3]2– |
2 |
Fe(H2O)3(OH)3 |
+ |
3 |
CO2 |
+ |
3 |
H2O |
[Al(H2O)6]3+ |
+ |
3 |
NaOH(aq) |
Al(H2O)3(OH)3 Al(OH)3 |
+ |
3 |
H2O |
+ |
3 |
Na+ |
[Al(H2O)6]3+ |
+ |
4 |
NaOH(aq) |
[Al(OH)4]– |
+ |
4 |
Na+ |
+ |
6 |
H2O |
Al(H2O)3(OH)3 Al(OH)3 |
+ |
NaOH(aq) |
[Al(OH)4]– |
+ |
Na+ |
+ |
3 |
H2O |
Al(H2O)3(OH)3 Al(OH)3 |
+ |
3 |
H+ |
[Al(H2O)6]3+ |
[Al(H2O)6]3+ |
+ |
3 |
NH3(aq) |
Al(H2O)3(OH)3 Al(OH)3 |
+ |
3 |
[NH4]+ |
[Al(H2O)6]3+ |
+ |
6 |
NH3(aq) |
Al(H2O)3(OH)3 Al(OH)3 |
+ |
3 |
[NH4]+ |
+ |
3 |
NH3 |
2 |
[Al(H2O)6]3+ |
+ |
3 |
[CO3]2– |
2 |
Al(H2O)3(OH)3 Al(OH)3 |
+ |
3 |
CO2 |
+ |
3 |
H2O |
[Cr(H2O)6]2+ |
+ |
3 |
NaOH(aq) |
Cr(H2O)3(OH)3 |
+ |
3 |
Na+ |
+ |
3 |
H2O |
[Cr(H2O)6]2+ |
+ |
6 |
NaOH(aq) |
[Cr(OH)6]3– |
+ |
6 |
H2O |
+ |
6 |
Na+ |
[Cr(H2O)6]2+ |
+ |
3 |
NH3(aq) |
Cr(H2O)3(OH)3 |
+ |
3 |
[NH4]+ |
2 |
[Cr(H2O)6]2+ |
+ |
3 |
[CO3]2– |
2 |
Cr(H2O)3(OH)3 |
+ |
3 |
CO2 |
+ |
3 |
H2O |
Cr(H2O)3(OH)3 |
+ |
6 |
NH3(aq) |
[Cr(NH3)6]2+ |
+ |
3 |
H2O |
+ |
3 |
[OH]– |
Fe(H2O)4(OH)2 |
+ |
2 |
H+ |
[Fe(H2O)6]2+ |
Co(H2O)4(OH)2 |
+ |
2 |
H+ |
[Co(H2O)6]2+ |
Cu(H2O)4(OH)2 |
+ |
2 |
H+ |
[Cu(H2O)6]2+ |
Fe(H2O)3(OH)3 |
+ |
3 |
H+ |
[Fe(H2O)6]3+ |
Cr(H2O)3(OH)3 |
+ |
3 |
H+ |
[Cr(H2O)6]2+ |
[Fe(H2O)6]3+ |
+ |
H2O |
[Co(NH3)6]2+ |
+ |
[H3O]+ |
Fe2(SO4)3 .5H2O |
+ |
7 |
H2O |
2 |
[Fe(H2O)6]3+ |
+ |
3 |
[SO4]2– |
Cr(H2O)3(OH)3 |
+ |
3 |
NaOH(aq) |
[Cr(OH)6]3– |
+ |
3 |
Na+ |
+ |
3 |
H2O |
[Cr(H2O)6]2+ |
+ |
H2O |
Cr(H2O)5(OH)]2+ |
+ |
[H3O]+ |
[Cr(H2O)6]2+ |
Cr(H2O)5(OH)]2+ |
+ |
H+ |
H+ |
+ |
[AlCl4]– |
AlCl3 |
+ |
HCl |
+ |
HBr |
+ |
Br– |
+ |
H+ |
+ |
Br– |
+ |
HBr |
3 |
Na2O |
+ |
2 |
H3PO4 |
2 |
Na3PO4 |
+ |
3 |
H2O |
Na2O |
+ |
2 |
H+ |
2 |
Na+ |
+ |
H2O |
2 |
NaF |
+ |
H2SO4 |
2 |
HF |
+ |
Na2SO4 |
2 |
NaBr |
+ |
H2SO4 |
2 |
HBr |
+ |
Na2SO4 |
4 |
NaBr |
+ |
3 |
H2SO4 |
2 |
HBr |
+ |
2 |
Na2SO4 |
+ |
Br2 |
+ |
SO2 |
+ |
2 |
H2O |
2 |
NaI |
+ |
H2SO4 |
2 |
HI |
+ |
Na2SO4 |
NaI |
+ |
H2SO4 |
HI |
+ |
I2 |
+ |
SO2 |
+ |
S |
+ |
H2S |
+ |
2 |
HCl(aq) |
+ |
2 |
Cl– |
+ |
NaOH(aq) |
+ |
Na+ |
+ |
H2O |
CH3COOH |
+ |
KOH |
+ |
H2O |
[CH3COO]– |
+ |
K+ |
+ |
H2O |
+ |
[OH]– |
+ |
H2O |
H2O |
H+ |
+ |
[OH]– |
BaCl2 |
+ |
H2SO4(aq) |
+ |
H2O |
BaSO4 |
+ |
2 |
HCl |
2 |
NH3 |
+ |
H2SO4 |
(NH4)2SO4 |
2 |
KF |
+ |
H2SO4 |
2 |
HF |
+ |
K2SO4 |
+ |
[OH]– |
+ |
HBr |
+ |
H+ |
HNO3 |
+ |
H2SO4 |
+ |
[HSO4]– |
HNO3|H2SO4 concentrated |
+ |
[HSO4]– |
RCOOH |
+ |
2 |
LiCH3 |
+ |
LiCH3 |
H2C=CH2 |
+ |
+ |
Li+ |
+ |
CH4 |
© Mark R. Leach 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com