
Interactions | Reactions | Processes
Classified as: Type 24 Lewis Acid/Base Complexation Chemistry
All Heavy metals are known to form some type of Type 24 pi-organometallic complex.
For more information look in the Chemogenesis webbook sections on Lewis acids and Lewis bases and the Lewis acid/base interaction matrix.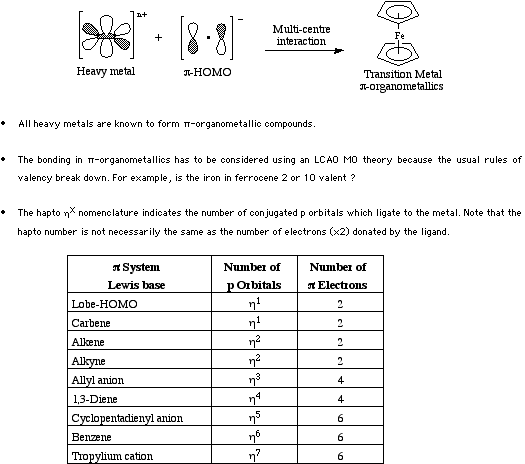
Ni |
+ |
4 |
CO |
Ni(CO)4 |
K2[PtCl4] |
+ |
H2C=CH2 |
+ |
KCl |
2 |
Pd2+ |
+ |
2 |
CH3–CH=CH2 |
+ |
4 |
Cl– |
2 |
Pd2+ |
+ |
2 |
+ |
2 |
Cl– |
Fe2+ |
+ |
+ |
3 |
CO |
Fe2+ |
+ |
2 |
+ |
CO |
2 |
Rh+ |
+ |
2 |
+ |
2 |
Cl– |
2 |
Rh+ |
+ |
2 |
+ |
2 |
Cl– |
Fe2+ |
+ |
2 |
Ni2+ |
+ |
2 |
Ti4+ |
+ |
2 |
+ |
2 |
Cl– |
Zr4+ |
+ |
2 |
+ |
H– |
+ |
Cl– |
Cr |
+ |
+ |
3 |
CO |
Cr |
+ |
+ |
3 |
CO |
Cr |
+ |
+ |
3 |
CO |
U4+ |
+ |
2 |
+ |
Fe |
+ |
5 |
CO |
Fe(CO)5 |
© Mark R. Leach 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com