
Interactions | Reactions | Processes
Classified as: Type 23 Lewis Acid/Base Complexation Chemistry
Very many Type 23 "transition metal coordination" complexes are known.
For more information look in the Chemogenesis webbook sections on Lewis acids and Lewis bases and the Lewis acid/base interaction matrix.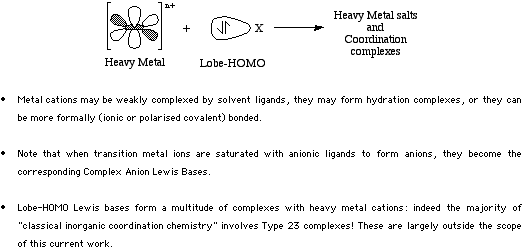
Pd |
+ |
4 |
[(C6H5)3P]4Pd |
Cr6+ |
+ |
3 |
O2– |
CrO3 |
Cu+ |
+ |
[CN]– |
CuCN |
Fe3+ |
+ |
3 |
Br– |
FeBr3 |
Fe3+ |
+ |
3 |
Cl– |
FeCl3 |
Pb4+ |
+ |
4 |
[CH3COO]– |
Pb(OCOCH3)4 |
Cu+ |
+ |
2 |
R– |
+ |
Li+ |
R2CuLi |
Hg2+ |
+ |
2 |
[CH3COO]– |
Hg(OCOCH3)2 |
Hg2+ |
+ |
O2– |
HgO |
Hg2+ |
+ |
[SO4]2– |
HgSO4 |
K+ |
+ |
[MnO4]– |
KMnO4 |
Sn2+ |
+ |
2 |
Cl– |
SnCl2 |
Zn2+ |
+ |
2 |
Br– |
ZnBr2 |
Bi3+ |
+ |
3 |
[NO3]– |
+ |
5 |
H2O |
Bi(NO3)2 |
Cd2+ |
+ |
2 |
Br– |
CdBr2 |
2 |
Ce3+ |
+ |
3 |
O2– |
Ce2O3 |
Ce4+ |
+ |
2 |
O2– |
CeO2 |
Cr2+ |
+ |
2 |
Cl– |
CrCl2 |
Cr3+ |
+ |
3 |
Cl– |
CrCl3 |
Co2+ |
+ |
[SO4]2– |
CoSO4 |
Cu2+ |
+ |
O2– |
CuO |
Fe2+ |
+ |
[CO3]2– |
FeCO3 |
Fe2+ |
+ |
[C2O4]2– |
FeC2O4 |
Pb2+ |
+ |
2 |
[CH3COO]– |
Pb(CH3COO)2 |
Ni2+ |
+ |
[CO3]2– |
NiCO3 |
Ni |
+ |
4 |
CO |
Ni(CO)4 |
Zr4+ |
+ |
4 |
Cl– |
ZrCl4 |
Ag+ |
+ |
Cl– |
AgCl |
Ag+ |
+ |
Br– |
AgBr |
Ag+ |
+ |
[BrO3]– |
AgBrO3 |
Ag+ |
+ |
[SCN]– |
AgSCN |
2 |
Ag+ |
+ |
[C2O4]2– |
AgC2O4 |
2 |
Ag+ |
+ |
[CrO4]2– |
Ag2CrO4 |
Ag+ |
+ |
I– |
AgI |
Ag+ |
+ |
[IO3]– |
AgIO3 |
3 |
Ag+ |
+ |
[PO4]3– |
Ag3PO4 |
2 |
Ag+ |
+ |
S2– |
Ag2S |
2 |
Ag+ |
+ |
[SO4]2– |
Ag2SO4 |
2 |
Bi3+ |
+ |
3 |
S2– |
Ba2S3 |
Cd2+ |
+ |
S2– |
CdS |
Co2+ |
+ |
2 |
[OH]– |
Co(OH)2 |
Co3+ |
+ |
3 |
[OH]– |
Co(OH)3 |
Co2+ |
+ |
S2– |
CoS |
Cu+ |
+ |
Br– |
CuBr |
Cu+ |
+ |
Cl– |
CuCl |
Cu+ |
+ |
I– |
CuI |
Cu2+ |
+ |
S2– |
2 |
Cu+ |
+ |
S2– |
Cu2S |
Cu+ |
+ |
[SCN]– |
CuSCN |
Fe2+ |
+ |
2 |
[OH]– |
Fe(OH)2 |
Fe3+ |
+ |
3 |
[OH]– |
Fe(OH)3 |
Fe2+ |
+ |
S2– |
FeS |
Hg23+ |
+ |
2 |
Br– |
Hg2Br2 |
Hg23+ |
+ |
2 |
Cl– |
Hg2Cl2 |
Hg23+ |
+ |
2 |
I– |
Hg2I2 |
Hg23+ |
+ |
S2– |
Hg2S |
Hg2+ |
+ |
S2– |
HgS |
Mg2+ |
+ |
2 |
[OH]– |
Mn(OH)2 |
Mg2+ |
+ |
S2– |
MnS |
Ni2+ |
+ |
2 |
[OH]– |
Ni(OH)2 |
Ni2+ |
+ |
S2– |
Pb2+ |
+ |
2 |
Br– |
PbBr2 |
Pb2+ |
+ |
2 |
Cl– |
PbCl2 |
Pb2+ |
+ |
[CO3]2– |
PbCO3 |
Pb2+ |
+ |
[CrO4]2– |
PbCrO4 |
Pb2+ |
+ |
2 |
F– |
PbF2 |
Pb2+ |
+ |
2 |
I– |
PbI2 |
3 |
Pb2+ |
+ |
2 |
[PO4]3– |
Pb3(PO4)2 |
Pb2+ |
+ |
S2– |
PbS |
Pb2+ |
+ |
[SO4]2– |
PbSO4 |
Tl+ |
+ |
Cl– |
TlCl |
Tl+ |
+ |
I– |
TlI |
2 |
Tl+ |
+ |
S2– |
Tl2S |
Zn2+ |
+ |
2 |
[OH]– |
Zn(OH)2 |
Zn2+ |
+ |
S2– |
ZnS |
Cu2+ |
+ |
2 |
[OH]– |
Cu(OH)2 |
Cu2+ |
+ |
2 |
[NO3]– |
+ |
6 |
H2O |
Cu(NO3)2 |
Pb2+ |
+ |
2 |
[NO3]– |
Pb(NO3)2 |
Ag+ |
+ |
[NO3]– |
AgNO3 |
Hg2+ |
+ |
2 |
Cl– |
HgCl2 |
Sn2+ |
+ |
O2– |
SnO |
Cu2+ |
+ |
[SO4]2– |
CuSO4 |
Zr4+ |
+ |
[SiO4]4– |
ZrSiO4 |
2 |
Zn2+ |
+ |
[SiO4]4– |
Zn2SiO4 |
Y3+ |
+ |
[PO4]3– |
YPO4 |
Pb2+ |
+ |
2 |
[MnO4]– |
Pb(MnO4)2 |
Mg2+ |
+ |
[WO4]2– |
MnWO4 |
Fe2+ |
+ |
[WO4]2– |
FeWO4 |
Zn2+ |
+ |
[CO3]2– |
ZnCO3 |
Mg2+ |
+ |
[CO3]2– |
MnCO3 |
U4+ |
+ |
2 |
O2– |
UO2 |
Ti4+ |
+ |
2 |
O2– |
TiO2 |
Sn4+ |
+ |
2 |
O2– |
Sn2O |
Fe2+ |
+ |
O2– |
FeO |
2 |
Fe3+ |
+ |
3 |
O2– |
Fe2O3 |
Fe2+ |
+ |
2 |
Fe3+ |
+ |
4 |
O2– |
FeO.Fe2O3 |
Zn2+ |
+ |
O2– |
ZnO |
W6+ |
+ |
3 |
O2– |
+ |
H2O |
WO3.H2O |
2 |
Cu+ |
+ |
O2– |
Cu2O |
Mo4+ |
+ |
2 |
S2– |
MoS2 |
2 |
Sb3+ |
+ |
3 |
S2– |
Sb2S3 |
2 |
As3+ |
+ |
3 |
S2– |
As2S3 |
+ |
Pt2+ |
+ |
H– |
+ |
2 |
(CH3CH2)3P |
+ |
Cl– |
Pd2+ |
+ |
2 |
Cl– |
PdCl2 |
Fe |
+ |
5 |
CO |
Fe(CO)5 |
4 |
Ag |
+ |
8 |
NaCN |
+ |
O2 |
+ |
2 |
H2O |
4 |
Na[Ag(CN)2] |
+ |
4 |
NaOH |
4 |
Au |
+ |
8 |
NaCN |
+ |
O2 |
+ |
2 |
H2O |
4 |
Na[Au(CN)2] |
+ |
4 |
NaOH |
Ag+ |
+ |
2 |
[CN]– |
[Ag(CN)2]– |
Au+ |
+ |
2 |
[CN]– |
[Au(CN)2]– |
Zn2+ |
+ |
2 |
Cl– |
ZnCl2 |
Pt2+ |
+ |
4 |
Cl– |
[PtCl4]2– |
Fe3O4 |
+ |
8 |
HCl |
FeCl2 |
+ |
2 |
FeCl3 |
+ |
4 |
H2O |
Zn2+ |
+ |
ZnS2O3 |
2 |
Co |
+ |
8 |
CO |
Co2(CO)8 |
Hg2+ |
+ |
2 |
[O=N=C]– |
Hg(ONC)2 |
Hg2+ |
+ |
2 |
[NO3]– |
Hg(NO3)2 |
Pb2+ |
+ |
2 |
[N3]– |
Pb(N3)2 |
Cu2+ |
+ |
4 |
Cl– |
[CuCl4]2– |
Cu2+ |
+ |
6 |
H2O |
[Cu(H2O)6]2+ |
CuSO4 |
+ |
6 |
H2O |
[Cu(H2O)6]2+ |
+ |
[SO4]2– |
Cu2+ |
+ |
[Cu(EDTA)]2– |
[Cu(H2O)6]2+ |
+ |
[Cu(EDTA)]2– |
+ |
6 |
H2O |
+ |
+ |
+ |
Fe2+ |
+ |
+ |
2 |
H+ |
Ag+ |
+ |
2 |
NH3 |
[Ag(NH3)2]+ |
Ag+ |
+ |
2 |
[Ag(S2O3)2]3– |
Ag+ |
+ |
2 |
[CN]– |
[Ag(CN)2]– |
Fe2+ |
+ |
6 |
H2O |
[Fe(H2O)6]2+ |
Fe3+ |
+ |
6 |
H2O |
[Fe(H2O)6]3+ |
Cr2+ |
+ |
6 |
H2O |
[Cr(H2O)6]2+ |
Cr3+ |
+ |
6 |
H2O |
[Cr(H2O)6]2+ |
[Cr(H2O)6]2+ |
+ |
6 |
NH3(aq) |
[Cr(NH3)6]2+ |
+ |
6 |
H2O |
Co2+ |
+ |
6 |
H2O |
[Co(H2O)6]2+ |
[Cu(H2O)6]2+ |
+ |
4 |
NH3(aq) |
[Cu(NH3)4(H2O)2]2+ |
+ |
4 |
H2O |
Co2+ |
+ |
4 |
Cl– |
[CoCl4]2– |
Cu2+ |
+ |
4 |
Cl– |
[CuCl4]2– |
[Cu(H2O)6]2+ |
+ |
4 |
Cl– |
[CuCl4]2– |
+ |
6 |
H2O |
[Co(H2O)6]2+ |
+ |
4 |
Cl– |
[CoCl4]2– |
+ |
6 |
H2O |
Fe2+ |
+ |
3 |
[Fe(H2O)6]2+ |
+ |
3 |
+ |
6 |
H2O |
+ |
+ |
6 |
+ |
3 |
+ |
6 |
Ni2+ |
+ |
3 |
[Ni(en)3]2+ |
[Fe(H2O)6]3+ |
+ |
3 |
[C2O4]2– |
[Fe(C2O4)3]3– |
+ |
6 |
H2O |
Fe3+ |
+ |
3 |
[C2O4]2– |
[Fe(C2O4)3]3– |
[Fe(H2O)6]3+ |
+ |
[SCN]– |
[Fe(H2O)5(SCN)]2+ |
+ |
H2O |
V3+ |
+ |
4 |
H2O |
+ |
2 |
Cl– |
[VCl2(H2O)4]+ |
V2+ |
+ |
6 |
H2O |
[V(H2O)6]2+ |
Mg2+ |
+ |
6 |
H2O |
[Mn(H2O)6]2+ |
AgBr |
+ |
2 |
+ |
hν |
[Ag(S2O3)2]3– |
+ |
Br– |
[Fe(H2O)6]2+ |
+ |
[SO4]2– |
+ |
H2O |
FeSO4.7H2O |
[Co(H2O)6]2+ |
+ |
2 |
Cl– |
CoCl2.6H2O |
[Fe(H2O)6]3+ |
+ |
3 |
[NO3]– |
+ |
3 |
H2O |
Fe(NO3)3 .9H2O |
[Al(H2O)6]3+ |
+ |
3 |
Cl– |
AlCl3.6H2O |
Fe2(SO4)3 .5H2O |
+ |
7 |
H2O |
2 |
[Fe(H2O)6]3+ |
+ |
3 |
[SO4]2– |
[Co(H2O)6]2+ |
+ |
4 |
HCl(conc) |
[CoCl4]2– |
+ |
4 |
H+ |
+ |
6 |
H2O |
AgCl |
+ |
2 |
NH3(aq) |
[Ag(NH3)2]+ |
+ |
Cl– |
[Ag(NH3)2]+ |
+ |
NaBr |
+ |
H2O |
AgBr |
+ |
2 |
NH3 |
+ |
Na+ |
AgBr |
+ |
2 |
Na2S2O3 |
+ |
H2O |
[Ag(S2O3)2]3– |
+ |
4 |
Na+ |
+ |
Br– |
AgBr |
+ |
2 |
KCN |
+ |
H2O |
[Ag(CN)2]– |
+ |
2 |
K+ |
+ |
Br– |
[Ag(CN)2]– |
+ |
e– |
+ |
K+ |
+ |
H2O |
Ag |
+ |
2 |
[CN]– |
+ |
K+ |
Co2+ |
+ |
3 |
[Co(en)3]2+ |
[Co(H2O)6]2+ |
+ |
[Co(EDTA)]3– |
+ |
6 |
H2O |
Co2+ |
+ |
[Co(EDTA)]3– |
CuCl2 |
+ |
2 |
HCl(conc) |
[CuCl4]2– |
+ |
2 |
H+ |
Cu+ |
+ |
4 |
Cl– |
[CuCl4]3– |
Ag+ |
+ |
2 |
Cl– |
[AgCl2]– |
AgBr |
+ |
2 |
.880 NH3 |
[Ag(NH3)2]+ |
+ |
Br– |
Ag+ |
+ |
[NO3]– |
+ |
Na+ |
+ |
Cl– |
+ |
H2O |
AgCl |
+ |
Na+ |
+ |
[NO3]– |
Ag+ |
+ |
[NO3]– |
+ |
Na+ |
+ |
Br– |
+ |
H2O |
AgBr |
+ |
Na+ |
+ |
[NO3]– |
Ag+ |
+ |
[NO3]– |
+ |
Na+ |
+ |
I– |
+ |
H2O |
AgI |
+ |
Na+ |
+ |
[NO3]– |
2 |
AgNO3 |
+ |
Na2CO3 |
+ |
H2O |
Ag2CO3 |
+ |
2 |
NaNO3 |
2 |
Ag+ |
+ |
2 |
[NO3]– |
+ |
2 |
Na+ |
+ |
[CO3]2– |
+ |
H2O |
Ag2CO3 |
+ |
2 |
Na+ |
+ |
2 |
[NO3]– |
2 |
Ag+ |
+ |
[CO3]2– |
Ag2CO3 |
Ag+ |
+ |
[NO3]– |
+ |
K+ |
+ |
Br– |
+ |
H2O |
AgBr |
+ |
K+ |
+ |
[NO3]– |
Ag+ |
+ |
Br– |
+ |
H2O |
AgBr |
Ag+ |
+ |
Cl– |
+ |
H2O |
AgCl |
AgBr |
+ |
2 |
[CN]– |
+ |
H2O |
[Ag(CN)2]– |
+ |
Br– |
Ag+ |
+ |
2 |
[CN]– |
[Ag(CN)2]– |
CaBr2 |
+ |
2 |
AgNO3 |
+ |
H2O |
2 |
AgBr |
+ |
Ca2+ |
+ |
2 |
[NO3]– |
[VO]2+ |
+ |
5 |
H2O |
[VO]2+ |
BaCl2 |
+ |
2 |
AgNO3 |
+ |
H2O |
2 |
AgCl |
+ |
Ba2+ |
+ |
2 |
[NO3]– |
2 |
Cu2+ |
+ |
4 |
I– |
+ |
H2O |
2 |
CuI |
+ |
I2 |
[CrO4]2– |
+ |
2 |
AgNO3 |
+ |
H2O |
Ag2CrO4 |
+ |
2 |
[NO3]– |
© Mark R. Leach 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com