
Interactions | Reactions | Processes
Classified as: Single Electron Transfer
As the name suggests, SET involves the transfer of a single electron from one species to another.
The species which donates the electron is oxidised an the species which receives the electron is reduced. If a neutral and spin paired molecular species receives an electron by SET (from a metal, electrode etc.) it becomes a radical anion. For more information look in the Chemogenesis webbook sections on redox chemistry and substitution, transfer, abstraction, displacement (STAD).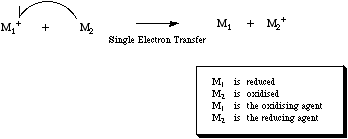
RCHO |
+ |
Zn(Hg) |
+ |
H+(aq) |
R–CH3 |
+ |
Zn2+ |
R–C≡C–R |
+ |
Li |
+ |
NH3 |
RCl |
+ |
Mg |
RMgCl |
RBr |
+ |
Mg |
RMgBr |
CH3I |
+ |
Mg |
CH3MgI |
+ |
Zn(Hg) |
+ |
H+(aq) |
+ |
2 |
Fe |
+ |
6 |
H+ |
+ |
2 |
H2O |
+ |
2 |
Fe3+ |
+ |
3 |
Sn |
+ |
6 |
H+ |
+ |
3 |
Sn2+ |
+ |
2 |
H2O |
RBr |
+ |
Li |
RLi |
+ |
LiBr |
RLi |
+ |
CuBr |
R2CuLi |
+ |
LiBr |
(C6H5)3CCl |
+ |
Ag |
(C6H5)3C• |
+ |
AgCl |
+ |
Na |
+ |
Na+ |
+ |
Fe3+ |
+ |
Fe2+ |
HOOH |
+ |
Fe2+ |
[H2O2]+• |
+ |
Fe3+ |
+ |
Na |
+ |
Na+ |
+ |
Isocitrate Dehydrogenase |
+ |
2 |
H• |
[H3]+ |
+ |
e– |
3 |
H• |
[H3]+ |
+ |
e– |
H2 |
+ |
H• |
© Mark R. Leach 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com