
Interactions | Reactions | Processes
Classified as: Type 16 Lewis Acid/Base Complexation Chemistry
As pi-HOMO systems tend to be highly susceptible to electrophilic attack, Lobe-LUMO Lewis acids initiate electrophilic substitution, electrophilic addition and addition-followed-by-elimination reactions.
For more information look in the Chemogenesis webbook sections on Lewis acids and Lewis bases and the Lewis acid/base interaction matrix.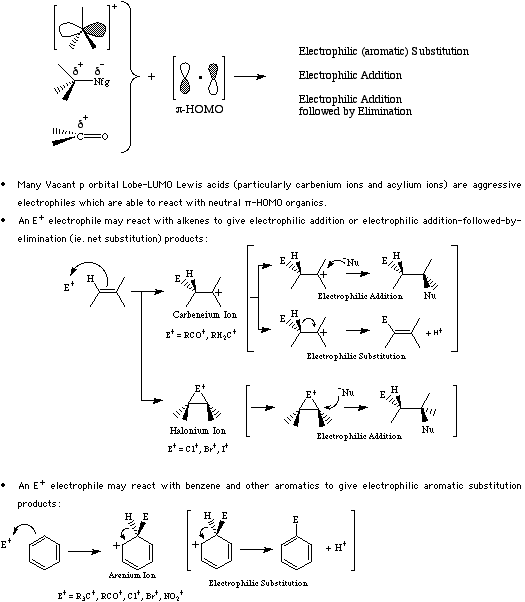
+ |
Cl2 |
+ |
Br2 |
+ |
HCl |
R–C≡C–R |
+ |
Cl2 |
CO2 |
+ |
CH2 |
+ |
B2H6 |
+ |
4 |
2 |
+ |
Br+ |
+ |
+ |
+ |
[CH3CO]+ |
CH3–CH=CH2 |
+ |
Br2 |
+ |
Br– |
+ |
[NO2]+ |
+ |
HNO3 |
+ |
H2SO4 |
+ |
H2O |
+ |
[HSO4]– |
© Mark R. Leach 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com