
Interactions | Reactions | Processes
Classified as: Type 15 Lewis Acid/Base Complexation Chemistry
All covalent sigma-bonds can be regarded as Type 15 complexes. Reactions involving Type 15 complexes occur either occur via 1st order sequential mechanisms (decomplexation followed by complexation) or via 1st order concerted mechanisms.
For more information look in the Chemogenesis webbook sections on Lewis acids and Lewis bases and the Lewis acid/base interaction matrix.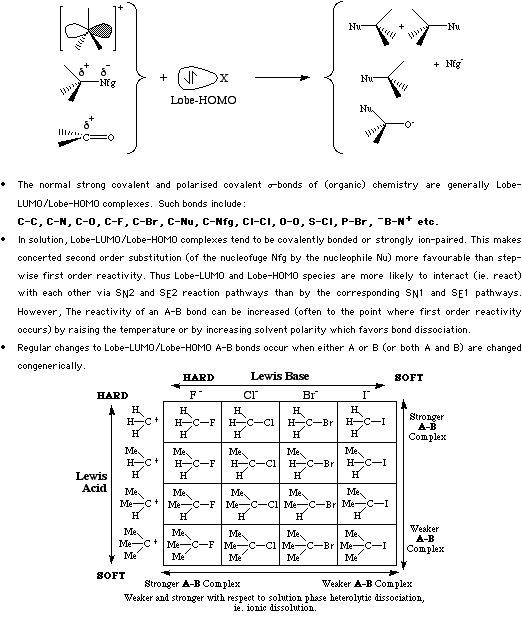
BF3 |
+ |
B2H6 |
+ |
2 |
2 |
+ |
Cl– |
R–O–CH2CH2Cl |
+ |
Cl– |
R2N–CH2CH2Cl |
+ |
Cl– |
R–S–CH2CH2Cl |
R+ |
+ |
Br– |
RBr |
R+ |
+ |
[CN]– |
R–NC |
+ |
[CN]– |
+ |
[CN]– |
Cl+ |
+ |
R– |
RCl |
R+ |
+ |
F– |
R–F |
R+ |
+ |
I– |
R–I |
R+ |
+ |
[H2N]– |
R–NH2 |
+ |
[H2N]– |
CO2 |
+ |
+ |
[CH3]– |
[(CH3)3C–O]– |
R+ |
+ |
–S–S– |
[RSS]– |
+ |
R–CH2O– |
R+ |
+ |
[N3]– |
R–N3 |
R+ |
+ |
RCOO– |
R+ |
+ |
[RSS]– |
R–S–S–R |
[CH3CO]+ |
+ |
[CH3COO]– |
(CH3CO)2CO |
Br+ |
+ |
Br– |
Br2 |
Cl+ |
+ |
Cl– |
Cl2 |
CH2 |
+ |
N2 |
CH2N2 |
Al3+ |
+ |
[CN]– |
+ |
2 |
[CH3CH2]– |
I+ |
+ |
I– |
I2 |
[CH3]+ |
+ |
I– |
CH3I |
[HO]+ |
+ |
[CH3COO]– |
CH3COOOH |
+ |
Br– |
2 |
+ |
[CO3]2– |
[CH3CO]+ |
+ |
RO– |
CH3COOR |
[CH3CO]+ |
+ |
[H2N]– |
CH3CONH2 |
[RCO]+ |
+ |
Cl– |
RCOCl |
2 |
Al3+ |
+ |
3 |
[SO4]2– |
Al2(SO4)3 |
Al3+ |
+ |
3 |
[OH]– |
Al(OH)3 |
Sb3+ |
+ |
3 |
Cl– |
SbCl3 |
Sb5+ |
+ |
5 |
Cl– |
SbCl5 |
As3+ |
+ |
3 |
F– |
AsF3 |
As5+ |
+ |
5 |
F– |
AsF5 |
Br+ |
+ |
Cl– |
BrCl |
[CH3]+ |
+ |
F– |
CH3F |
[CH3CH2]+ |
+ |
F– |
+ |
F– |
[CH3]+ |
+ |
Cl– |
CH3Cl |
[CH3CH2]+ |
+ |
Cl– |
CH3CH2Cl |
+ |
Cl– |
+ |
F– |
+ |
Cl– |
[CH3]+ |
+ |
Br– |
CH3Br |
[CH3CH2]+ |
+ |
Br– |
CH3CH2Br |
+ |
Br– |
(CH3)2CHBr |
[CH3CH2]+ |
+ |
I– |
CH3CH2I |
+ |
I– |
(CH3)2CHI |
+ |
I– |
(CH3)3CI |
RCHO |
+ |
[CN]– |
R–C–H |
+ |
Br+ |
+ |
+ |
Cl– |
+ |
Br– |
+ |
I– |
Cl+ |
+ |
[RCO]+ |
+ |
[RCO]+ |
+ |
+ |
Cl+ |
Br+ |
+ |
FeBr3 |
+ |
Br– |
[FeBr4]– |
+ |
I+ |
[NO2]+ |
+ |
[HSO4]– |
+ |
H2O |
HNO3|H2SO4 concentrated |
R+ |
+ |
+ |
[CN]– |
[RCO]+ |
+ |
RCOO– |
(RCOO)2O |
[CH3]+ |
+ |
RCOO– |
R+ |
+ |
R–Nu |
Br+ |
+ |
[RCH2CH2]+ |
+ |
[H2N]– |
RCH2NH2 |
R+ |
+ |
R– |
R–R |
[RCO]+ |
+ |
R+ |
+ |
Br+ |
+ |
[CH3]+ |
+ |
[R–S]– |
R–S–CH3 |
[RCH2CH2]+ |
+ |
[(C6H5)3P–CH2R]+ |
[CH3CH2]+ |
+ |
[PO4]3– |
(CH3CH2O)3P=O |
[CH3CO]+ |
+ |
[CH3CH2]– |
[CH3CO]+ |
+ |
[(CH3)3C]– |
Br+ |
+ |
R– |
RBr |
CH2 |
+ |
+ |
[CN]– |
R+ |
+ |
Cl– |
RCl |
I+ |
+ |
R– |
R–I |
[CH3]+ |
+ |
[CN]– |
CH3CN |
R+ |
+ |
[CN]– |
R–CN |
[CH3CH2]+ |
+ |
[H2N]– |
CH3CH2NH2 |
CH3CHO |
+ |
[CH3]– |
[(CH3)2CH–O]– |
+ |
[CH3]– |
CH3CHO |
[CH3CO]+ |
+ |
[CH3]– |
[CH3CO]+ |
+ |
[OH]– |
CH3COOH |
+ |
[OH]– |
(CH3)3COH |
[RCH2CH2]+ |
+ |
[OH]– |
RCH2OH |
[RCO]+ |
+ |
[H2N]– |
R+ |
+ |
[HS]– |
R–SH |
R+ |
+ |
[HTe]– |
R–TeH |
R+ |
+ |
NH3 |
[R–NH3]+ |
R+ |
+ |
HBr |
[R–BrH]+ |
[CH3CO]+ |
+ |
R+ |
+ |
HCl |
[R–ClH]+ |
R+ |
+ |
HF |
[R–FH]+ |
R+ |
+ |
HI |
[R–IH]+ |
+ |
NH3 |
R+ |
+ |
H2S |
[R–SH2]+ |
R+ |
+ |
H2Te |
[R–TeH2]+ |
[RCO]+ |
+ |
[OH]– |
RCOOH |
R+ |
+ |
[HOO]– |
ROOH |
+ |
H2O |
+ |
H2O |
+ |
R– |
R+ |
+ |
[OH]– |
R–OH |
[RCO]+ |
+ |
RO– |
[R–S]+ |
+ |
[R–S]– |
R–S–S–R |
I+ |
+ |
[CH3]– |
CH3I |
Br+ |
+ |
Cl+ |
+ |
[CH3]– |
CH3Cl |
Cl+ |
+ |
[CH3CH2]– |
CH3CH2Cl |
Cl+ |
+ |
Cl+ |
+ |
[(CH3)3C]– |
Br+ |
+ |
[CH3]– |
CH3Br |
Br+ |
+ |
[CH3CH2]– |
CH3CH2Br |
Br+ |
+ |
(CH3)2CHBr |
I+ |
+ |
[CH3CH2]– |
CH3CH2I |
I+ |
+ |
(CH3)2CHI |
I+ |
+ |
[(CH3)3C]– |
(CH3)3CI |
[RCO]+ |
+ |
[CH3–O]– |
[RCO]+ |
+ |
R– |
[R3C]+ |
+ |
[OH]– |
R3COH |
+ |
RCOO– |
+ |
RO– |
[CH3CO]+ |
+ |
[R–S]+ |
+ |
[CH3]– |
R–S–CH3 |
Br+ |
+ |
R–CH2–Br |
[NO]+ |
+ |
[OH]– |
HNO2 |
+ |
+ |
+ |
[CH3CH2]+ |
+ |
H2O |
CH3CH2OH |
+ |
H+ |
[RCH2CH2]+ |
+ |
H2O |
R–CH2CH2OH |
+ |
H+ |
[R2CH]+ |
+ |
H2O |
R2CHOH |
+ |
H+ |
[R3C]+ |
+ |
H2O |
R3COH |
+ |
H+ |
B2H6 |
+ |
2 |
(CH3)2S |
2 |
(CH3)2S→BH3 |
Br+ |
+ |
Br– |
Br2(g) |
2 |
Al3+ |
+ |
3 |
O2– |
Al2O3 |
+ |
+ |
R+ |
+ |
R–Nfg |
[R3C]+ |
+ |
(R)3C–Nfg |
[R2CH]+ |
+ |
(R)2CH–Nfg |
[RCH2CH2]+ |
+ |
RCH2–Nfg |
[CH3]+ |
+ |
CH3–Nfg |
+ |
+ |
[RCO]+ |
+ |
+ |
RMgX |
+ |
SO2 |
RMgX |
+ |
SO2 |
[R–SO2]– |
R+ |
+ |
[NCO]– |
R–O–C≡N |
R+ |
+ |
[SCN]– |
R–N=C=S |
+ |
AlCl3 |
+ |
[AlCl4]– |
CO2 |
+ |
2 |
KOH |
K2CO3 |
+ |
H2O |
CO2 |
+ |
2 |
NaOH |
Na2CO3 |
+ |
H2O |
CO2 |
+ |
[OH]– |
[HCO3]– |
CO2 |
+ |
2 |
[OH]– |
[CO3]2– |
+ |
H2O |
[CO3]2– |
+ |
CO2 |
+ |
H2O |
+ |
H2O |
2 |
[HCO3]– |
SiF4 |
+ |
2 |
F– |
[SiF6]2– |
N2O4 |
[NO]+ |
+ |
[NO3]– |
[NO2]+ |
+ |
[NO3]– |
N2O5 |
SO3 |
+ |
H2S |
+ |
Cl+ |
+ |
[ClO4]– |
Cl+ [ClO4]– |
SbF5 |
+ |
HSO3F |
ROH |
+ |
SO3 |
BF3 |
+ |
ClF3 |
[ClF2]+ |
+ |
[BF4]– |
SbF5 |
+ |
ClF3 |
[ClF2]+ |
+ |
[SbF6]– |
BrF5 |
+ |
CsF |
Cs+ |
+ |
[BrF6]– |
IF5 |
+ |
KF |
K+ |
+ |
[IF6]– |
IF7 |
+ |
CsF |
Cs+ |
+ |
[IF8]– |
I–Cl |
+ |
KCl |
K+ |
+ |
[ICl2]– |
I2 |
+ |
I– |
[I3]– |
I–Cl |
+ |
KI |
K+ |
+ |
[I2Cl]– |
I2 |
+ |
I–Cl |
+ |
AlCl3 |
[I3]+ |
+ |
[AlCl4]– |
Cl2 |
+ |
ClF3 |
+ |
AsF5 |
[Cl3]+ |
+ |
[AsF6]– |
+ |
F2 |
Br2 |
+ |
BrF3 |
+ |
AsF5 |
[Br3]+ |
+ |
[AsF6]– |
+ |
F2 |
+ |
[OH]– |
[NO]+ |
+ |
H+ |
+ |
[SO4]2– |
NOHSO4 |
CH3COCl |
+ |
AlCl3 |
[CH3CO]+ |
+ |
[AlCl4]– |
Al(OH)3 |
+ |
NaOH |
[Al(OH)4]– |
+ |
Na+ |
Al(OH)3 |
+ |
NaOH(aq) |
[Al(OH)4]– |
+ |
Na+ |
+ |
Br– |
(CH3)3CBr |
Be(OH)2 |
+ |
2 |
[OH]– |
+ |
H2O |
[Be(OH)4]2– |
Be(OH)2 |
+ |
2 |
NaOH(aq) |
2 |
Na+ |
+ |
[Be(OH)4]2– |
AlCl3 |
+ |
Cl– |
[AlCl4]– |
+ |
KCN |
+ |
KBr |
+ |
Br– |
+ |
Br– |
+ |
Br– |
(CH3)2CHBr |
[CH3CH2]+ |
+ |
H2O |
[CH3CH2]+ |
+ |
Br– |
CH3CH2Br |
[NO2]+ |
+ |
H2O |
2 |
AlCl3 |
Al2Cl6 |
© Mark R. Leach 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com