
Interactions | Reactions | Processes
Classified as: Complexation or Coupling
Complexation is the process where by two species join together to form a single species.
The joining process may proceed via LUMO/HOMO Lewis acid/Lewis base, SOMO/SOMO radical/radical or steric Guest/Host types of interaction. Complexation processes are bimolecular in the formation direction and unimolecular in the decomplexation direction. For more information look in the Chemogenesis webbook sections on complexation, radical chemistry, addition reactions, Lewis acid/base interaction matrix. and pericyclic reaction chemistry.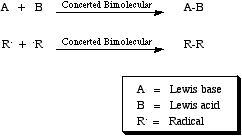
BF3 |
+ |
B2H6 |
+ |
2 |
2 |
2 |
BH3 |
B2H6 |
A+ Cationic Lewis acid |
+ |
Base– Proton abstractor |
A–B Neutral complex |
A+ Cationic Lewis acid |
+ |
Base: Proton abstractor |
[A–B]+ Cationic complex |
A Lewis acid |
+ |
Base– Proton abstractor |
[A–B]– Anionic complex |
A Lewis acid |
+ |
Base: Proton abstractor |
[A←B] Dative complex |
+ |
KMnO4 |
+ |
+ |
[MnO4]– |
+ |
HBF4(aq) |
+ |
H2O |
+ |
+ |
[BF4]– |
Li+ |
+ |
+ |
+ |
+ |
KI |
+ |
I2 |
+ |
H2O |
K+ |
+ |
[I3]– |
K+ |
+ |
3 |
+ |
H+ |
Li+ |
+ |
+ |
H+ |
Li+ |
+ |
2 |
+ |
H+ |
[H3O]+ |
+ |
H2O |
[H3O]+ |
+ |
3 |
H2O |
4 |
Ag |
+ |
8 |
NaCN |
+ |
O2 |
+ |
2 |
H2O |
4 |
Na[Ag(CN)2] |
+ |
4 |
NaOH |
4 |
Au |
+ |
8 |
NaCN |
+ |
O2 |
+ |
2 |
H2O |
4 |
Na[Au(CN)2] |
+ |
4 |
NaOH |
Ag+ |
+ |
2 |
[CN]– |
[Ag(CN)2]– |
Au+ |
+ |
2 |
[CN]– |
[Au(CN)2]– |
SiF4 |
+ |
2 |
F– |
[SiF6]2– |
GeF4 |
+ |
2 |
F– |
[GeF6]2– |
GeCl4 |
+ |
2 |
Cl– |
[GeCl6]2– |
SnF4 |
+ |
2 |
F– |
[SnF6]2– |
SnCl4 |
+ |
2 |
Cl– |
[SnCl6]2– |
PbF4 |
+ |
2 |
F– |
[PbF6]2– |
PbCl4 |
+ |
2 |
Cl– |
[PbCl6]2– |
NO |
+ |
NO2 |
N2O3 |
KF |
+ |
HF |
K[HF2] |
I2 |
+ |
I– |
[I3]– |
2 |
H2C=C=O |
O3 |
+ |
O |
2 |
O2 |
O2 |
+ |
O |
+ |
m |
O3 |
+ |
m* |
H2C=C=O |
+ |
CH3COOH |
(CH3CO)2CO |
Cu2+ |
+ |
6 |
H2O |
[Cu(H2O)6]2+ |
CuSO4 |
+ |
6 |
H2O |
[Cu(H2O)6]2+ |
+ |
[SO4]2– |
Cu2+ |
+ |
[Cu(EDTA)]2– |
[Cu(H2O)6]2+ |
+ |
[Cu(EDTA)]2– |
+ |
6 |
H2O |
+ |
+ |
+ |
Fe2+ |
+ |
+ |
2 |
H+ |
Ag+ |
+ |
2 |
NH3 |
[Ag(NH3)2]+ |
Ag+ |
+ |
2 |
[Ag(S2O3)2]3– |
Ag+ |
+ |
2 |
[CN]– |
[Ag(CN)2]– |
Fe2+ |
+ |
6 |
H2O |
[Fe(H2O)6]2+ |
Fe3+ |
+ |
6 |
H2O |
[Fe(H2O)6]3+ |
Cr2+ |
+ |
6 |
H2O |
[Cr(H2O)6]2+ |
Cr3+ |
+ |
6 |
H2O |
[Cr(H2O)6]2+ |
Co2+ |
+ |
6 |
H2O |
[Co(H2O)6]2+ |
Co2+ |
+ |
4 |
Cl– |
[CoCl4]2– |
Cu2+ |
+ |
4 |
Cl– |
[CuCl4]2– |
Fe2+ |
+ |
3 |
2 |
[CrO4]2– |
+ |
2 |
H+ |
[Cr2O7]2– |
+ |
H2O |
Al(OH)3 |
+ |
NaOH(aq) |
[Al(OH)4]– |
+ |
Na+ |
[Co(H2O)6]2+ |
+ |
[Co(EDTA)]3– |
+ |
6 |
H2O |
+ |
Br– |
(CH3)3CBr |
AgBr |
+ |
2 |
.880 NH3 |
[Ag(NH3)2]+ |
+ |
Br– |
+ |
Br– |
CH3–CH=CH2 |
+ |
Br2 |
+ |
Br– |
+ |
Br– |
+ |
Br– |
(CH3)2CHBr |
Cl3C• |
+ |
Cl• |
CCl4 |
[CH3CH2]+ |
+ |
Br– |
CH3CH2Br |
AgBr |
+ |
2 |
[CN]– |
+ |
H2O |
[Ag(CN)2]– |
+ |
Br– |
Ag+ |
+ |
2 |
[CN]– |
[Ag(CN)2]– |
+ |
[NO2]+ |
+ |
HNO3 |
+ |
H2SO4 |
+ |
H2O |
+ |
[HSO4]– |
2 |
AlCl3 |
Al2Cl6 |
[VO]2+ |
+ |
5 |
H2O |
[VO]2+ |
2 |
+ |
2 |
Na |
+ |
2 |
NaBr |
2 |
CH3COONa |
+ |
2 |
H2O |
CH3CH3 |
+ |
2 |
CO2 |
+ |
H2 |
+ |
2 |
NaOH |
© Mark R. Leach 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com